Connexin Hemichannel Activation by S-Nitrosoglutathione Synergizes Strongly with Photodynamic Therapy Potentiating Anti-Tumor Bystander Killing
- PMID: 34680212
- PMCID: PMC8533914
- DOI: 10.3390/cancers13205062
Connexin Hemichannel Activation by S-Nitrosoglutathione Synergizes Strongly with Photodynamic Therapy Potentiating Anti-Tumor Bystander Killing
Abstract
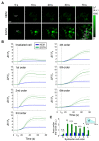
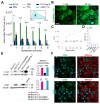
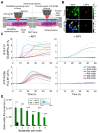
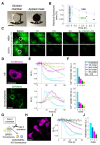
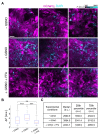
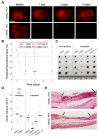
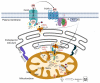
In this study, we used B16-F10 cells grown in the dorsal skinfold chamber (DSC) preparation that allowed us to gain optical access to the processes triggered by photodynamic therapy (PDT). Partial irradiation of a photosensitized melanoma triggered cell death in non-irradiated tumor cells. Multiphoton intravital microscopy with genetically encoded fluorescence indicators revealed that bystander cell death was mediated by paracrine signaling due to adenosine triphosphate (ATP) release from connexin (Cx) hemichannels (HCs). Intercellular calcium (Ca2+) waves propagated from irradiated to bystander cells promoting intracellular Ca2+ transfer from the endoplasmic reticulum (ER) to mitochondria and rapid activation of apoptotic pathways. Combination treatment with S-nitrosoglutathione (GSNO), an endogenous nitric oxide (NO) donor that biases HCs towards the open state, greatly potentiated anti-tumor bystander killing via enhanced Ca2+ signaling, leading to a significant reduction of post-irradiation tumor mass. Our results demonstrate that HCs can be exploited to dramatically increase cytotoxic bystander effects and reveal a previously unappreciated role for HCs in tumor eradication promoted by PDT.
Keywords: calcium signaling; nitric oxide; photosensitization; purinergic signaling.
Conflict of interest statement
The authors declare no conflict of interests. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
Similar articles
-
Photosensitizer Activation Drives Apoptosis by Interorganellar Ca2+ Transfer and Superoxide Production in Bystander Cancer Cells.Cells. 2019 Sep 29;8(10):1175. doi: 10.3390/cells8101175. Cells. 2019. PMID: 31569545 Free PMC article.
-
Cx43 channels and signaling via IP3/Ca2+, ATP, and ROS/NO propagate radiation-induced DNA damage to non-irradiated brain microvascular endothelial cells.Cell Death Dis. 2020 Mar 18;11(3):194. doi: 10.1038/s41419-020-2392-5. Cell Death Dis. 2020. PMID: 32188841 Free PMC article.
-
Intercellular calcium signaling in astrocytes via ATP release through connexin hemichannels.J Biol Chem. 2002 Mar 22;277(12):10482-8. doi: 10.1074/jbc.M109902200. Epub 2002 Jan 14. J Biol Chem. 2002. PMID: 11790776
-
Purinergic Signaling Is a Novel Mechanism of the Cellular Response to Ionizing Radiation.Biol Pharm Bull. 2015;38(7):951-9. doi: 10.1248/bpb.b15-00062. Biol Pharm Bull. 2015. PMID: 26133701 Review.
-
Calcium, oxidative stress and connexin channels, a harmonious orchestra directing the response to radiotherapy treatment?Biochim Biophys Acta Mol Cell Res. 2017 Jun;1864(6):1099-1120. doi: 10.1016/j.bbamcr.2017.02.007. Epub 2017 Feb 11. Biochim Biophys Acta Mol Cell Res. 2017. PMID: 28193563 Review.
Cited by
-
Commercially derived versatile optical architecture for two-photon STED, wavelength mixing and label-free microscopy.Biomed Opt Express. 2022 Feb 14;13(3):1410-1429. doi: 10.1364/BOE.444525. eCollection 2022 Mar 1. Biomed Opt Express. 2022. PMID: 35414982 Free PMC article.
-
The Multifaceted Role of Connexins in Tumor Microenvironment Initiation and Maintenance.Biology (Basel). 2023 Jan 28;12(2):204. doi: 10.3390/biology12020204. Biology (Basel). 2023. PMID: 36829482 Free PMC article. Review.
-
Enhanced Photodynamic Therapy Synergizing with Inhibition of Tumor Neutrophil Ferroptosis Boosts Anti-PD-1 Therapy of Gastric Cancer.Adv Sci (Weinh). 2024 Mar;11(12):e2307870. doi: 10.1002/advs.202307870. Epub 2024 Jan 17. Adv Sci (Weinh). 2024. PMID: 38233204 Free PMC article.
-
A Quantitative Assay for Ca2+ Uptake through Normal and Pathological Hemichannels.Int J Mol Sci. 2022 Jun 30;23(13):7337. doi: 10.3390/ijms23137337. Int J Mol Sci. 2022. PMID: 35806342 Free PMC article.
References
-
- Dabrowska A., Goś M., Janik P. Bystander effect induced by photodynamically or heat-injured ovarian carcinoma cells (OVP10) In Vitro. Med. Sci. Monit. 2005;11:324. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

