Novel Non-Immunologic Agents for Relapsed and Refractory Multiple Myeloma: A Review Article
- PMID: 34680358
- PMCID: PMC8534104
- DOI: 10.3390/cancers13205210
Novel Non-Immunologic Agents for Relapsed and Refractory Multiple Myeloma: A Review Article
Abstract
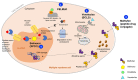
Novel treatments are needed to address the lack of options for patients with relapsed or refractory multiple myeloma. Even though immunotherapy-based treatments have revolutionized the field in recent years, offering new opportunities for patients, there is still no curative therapy. Thus, non-immunologic agents, which have proven effective for decades, are still central to the treatment of multiple myeloma, especially for advanced disease. Building on their efficacy in myeloma, the development of proteasome inhibitors and immunomodulatory drugs has been pursued, and has led to the emergence of a novel generation of agents (e.g., carfilzomib, ixazomib, pomalidomide). The use of alkylating agents is decreasing in most treatment regimens, but melflufen, a peptide-conjugated alkylator with a completely new mechanism of action, offers interesting opportunities. Moreover, with the identification of novel targets, new drug classes have entered the myeloma armamentarium, such as XPO1 inhibitors (selinexor), HDAC inhibitors (panobinostat), and anti-BCL-2 agents (venetoclax). New pathways are still being explored, especially the possibility of a mutation-driven strategy, as biomarkers and targeted treatments are increasing. Though multiple myeloma is still considered incurable, the treatment options are expanding and are progressively becoming more diverse, largely because of the continuous development of non-immunologic agents.
Keywords: HDAC inhibitors; IMiDs; XPO1 inhibitors; anti BCL-2; anti MCL-1; multiple myeloma; non-immunologic agents; peptide–drug conjugates; proteasome inhibitors; relapsed or refractory.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Kaufman J.L., Usmani S.Z., San-Miguel J., Bahlis N., White D.J., Benboubker L., Cook G., Leiba M., Ho P.J., Kim K., et al. Four-Year Follow-up of the Phase 3 Pollux Study of Daratumumab Plus Lenalidomide and Dexamethasone (D-Rd) Versus Lenalidomide and Dexamethasone (Rd) Alone in Relapsed or Refractory Multiple Myeloma (RRMM) Blood. 2019;134:1866. doi: 10.1182/blood-2019-123483. - DOI
-
- Bahlis N., Facon T., Usmani S.Z., Kumar S.K., Plesner T., Orlowski R.Z., Touzeau C., Basu S., Nahi H., Hulin C., et al. Daratumumab Plus Lenalidomide and Dexamethasone (D-Rd) Versus Lenalidomide and Dexamethasone (Rd) in Patients with Newly Diagnosed Multiple Myeloma (NDMM) Ineligible for Transplant: Updated Analysis of Maia. Blood. 2019;134:1875. doi: 10.1182/blood-2019-123426. - DOI
-
- Moreau P., Attal M., Hulin C., Arnulf B., Belhadj K., Benboubker L., Béné M.C., Broijl A., Caillon H., Caillot D., et al. Bortezomib, thalidomide, and dexamethasone with or without daratumumab before and after autologous stem-cell transplantation for newly diagnosed multiple myeloma (CASSIOPEIA): A randomised, open-label, phase 3 study. Lancet. 2019;394:29–38. doi: 10.1016/S0140-6736(19)31240-1. - DOI - PubMed
-
- Krejcik J., Frerichs K.A., Nijhof I.S., Van Kessel B., Van Velzen J.F., Bloem A.C., Broekmans M.E.C., Zweegman S., Van Meerloo J., Musters R.J.P., et al. Monocytes and granulocytes reduce CD38 expression levels on myeloma cells in patients treated with daratumumab. Clin. Cancer Res. 2017;23:7498–7511. doi: 10.1158/1078-0432.CCR-17-2027. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

