Metal Nanoparticles in Laser Bioprinting
- PMID: 34685024
- PMCID: PMC8539905
- DOI: 10.3390/nano11102584
Metal Nanoparticles in Laser Bioprinting
Abstract
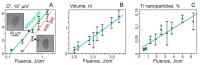
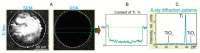
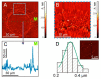
Laser bioprinting is a promising method for applications in biotechnology, tissue engineering, and regenerative medicine. It is based on a microdroplet transfer from a donor slide induced by laser pulse heating of a thin metal absorption film covered with a layer of hydrogel containing living cells (bioink). Due to the presence of the metal absorption layer, some debris in the form of metal nanoparticles is printed together with bioink microdroplets. In this article, experimental investigations of the amount of metal nanoparticles formed during the laser bioprinting process and transported in bioink microdroplets are performed. As metal absorption layers, Ti films with the thickness in the range of 25-400 nm, produced by magnetron spattering, were applied. Dependences of the volume of bioink microdroplets and the amount of Ti nanoparticles within them on the laser pulse fluence were obtained. It has been experimentally found that practically all nanoparticles remain in the hydrogel layer on the donor slide during bioprinting, with only a small fraction of them transferred within the microdroplet (0.5% to 2.5%). These results are very important for applications of laser bioprinting since the transferred metal nanoparticles can potentially affect living systems. The good news is that the amount of such nanoparticles is very low to produce any negative effect on the printed cells.
Keywords: Ti nanoparticles; gel microdroplets; laser bioprinting.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
-
- Serra P., Piqué A. Laser-Induced Forward Transfer: Fundamentals and Applications. Adv. Mater. Technol. 2019;4:1800099. doi: 10.1002/admt.201800099. - DOI
-
- Guillotin B., Souquet A., Catros S., Duocastella M., Pippenger B., Bellance S., Bareille R., Rémy M., Bordenave L., Amédée J., et al. Laser assisted bioprinting of engineered tissue with high cell density and microscale organization. Biomaterials. 2010;31:7250–7256. doi: 10.1016/j.biomaterials.2010.05.055. - DOI - PubMed
-
- Keriquel V., Oliveira H., Rémy M., Ziane S., Delmond S., Rousseau B., Rey S., Catros S., Amédée J., Guillemot F., et al. In situ printing of mesenchymal stromal cells, by laser-assisted bioprinting, for in vivo bone regeneration applications. Sci. Rep. 2017;7:1–10. doi: 10.1038/s41598-017-01914-x. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

