Acute cardiotoxicity after initiation of the novel tyrosine kinase inhibitor gilteritinib for acute myeloid leukemia
- PMID: 34686212
- PMCID: PMC8531894
- DOI: 10.1186/s40959-021-00122-x
Acute cardiotoxicity after initiation of the novel tyrosine kinase inhibitor gilteritinib for acute myeloid leukemia
Abstract
Background: Gilteritinib is a novel FMS-like tyrosine kinase 3 inhibitor recently approved by the United States Food and Drug Administration in 2018 for relapsed or refractory acute myeloid leukemia. However, gilteritinib may be associated with underrecognized cardiotoxicities.
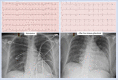
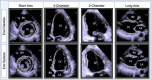
Case presentation: This case describes a patient with a history significant for hyperlipidemia who was diagnosed with relapsed acute myeloid leukemia. After four doses of gilteritinib monotherapy, she abruptly developed acute systolic heart failure with global hypokinesis and septal wall motion abnormalities. Two days after discontinuation, cardiac magnetic resonance imaging showed partial recovery of her left ventricular ejection fraction as well as myocardial edema and non-ischemic fibrosis suggestive of inflammatory cardiomyopathy. She underwent intravenous diuresis and eventually started guideline-directed heart failure therapy. Follow-up cardiac magnetic resonance imaging five months later showed improved ejection fraction with mild non-ischemic fibrosis and resolution of myocardial edema and inflammation. She later received an allogeneic stem cell transplant from a matched unrelated donor.
Conclusions: Gilteritinib may be associated with early cardiotoxicities, including non-ischemic cardiomyopathy and myocarditis. Cardiac magnetic resonance imaging can be an important modality to help differentiate or diagnose early cardiotoxicities associated with novel targeted therapies.
Keywords: Acute myeloid leukemia; Cardiac MRI; Cardio-oncology; FLT3-mutation; Gilteritinib.
© 2021. The Author(s).
Conflict of interest statement
All authors declare no conflicts of interests or competing interests in relation to the work presented in this manuscript.
Figures

References
-
- Thavendiranathan P, Zhang L, Zafar A, Drobni ZD, Mahmood SS, Cabral M, Awadalla M, Nohria A, Zlotoff DA, Thuny F, et al. Myocardial T1 and T2 Mapping by Magnetic Resonance in Patients With Immune Checkpoint Inhibitor-Associated Myocarditis. J Am Coll Cardiol. 2021;77(12):1503–1516. doi: 10.1016/j.jacc.2021.01.050. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
