IL-36α Enhances Host Defense against Pseudomonas aeruginosa Keratitis in C57BL/6 Mouse Corneas
- PMID: 34686582
- PMCID: PMC8612993
- DOI: 10.4049/jimmunol.2001246
IL-36α Enhances Host Defense against Pseudomonas aeruginosa Keratitis in C57BL/6 Mouse Corneas
Abstract
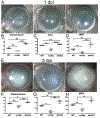
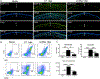
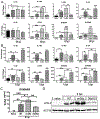
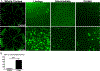
The IL-36 cytokines are known to play various roles in mediating the immune response to infection in a tissue- and pathogen-dependent manner. The present study seeks to investigate the role of IL-36R signaling in C57BL/6 mouse corneas in response to Pseudomonas aeruginosa infection. IL-36α-/-, IL-36γ-/-, and IL-36R-/- mice had significantly more severe keratitis than wild-type mice. At six hours postinfection, IL-36α pretreatment augmented P. aeruginosa-induced expression of IL-1Ra, IL-36γ, LCN2, and S100A8/A9. At one day postinfection, exogenous IL-36α suppressed, whereas IL-36α deficiency promoted, the expression of IL-1β. At three days postinfection, exogenous IL-36α suppressed Th1 but promoted Th2 immune response. IL-36α stimulated the infiltration of IL-22-expressing immune cells, and IL-22 neutralization resulted in more severe keratitis. IL-36α alone stimulated dendritic cell infiltration in B6 mouse corneas. Taken together, our study suggests that IL-36R signaling plays a protective role in the pathogenesis of P. aeruginosa keratitis by promoting the innate immune defense, Th2, and/or Th22/IL-22 immune responses. Exogenous IL-36α might be a potential therapy for improving the outcome of P. aeruginosa keratitis.
Copyright © 2021 by The American Association of Immunologists, Inc.
Figures




References
-
- Cheng KH, Leung SL, Hoekman HW, Beekhuis WH, Mulder PG, Geerards AJ, and Kijlstra A. 1999. Incidence of contact-lens-associated microbial keratitis and its related morbidity. Lancet 354: 181–185. - PubMed
-
- Slusher MM, Myrvik QN, Lewis JC, and Gristina AG. 1987. Extended-wear lenses, biofilm, and bacterial adhesion. Arch Ophthalmol 105: 110–115. - PubMed
-
- Guembel HO, and Ohrloff C. 1997. Opportunistic infections of the eye in immunocompromised patients. Ophthalmologica 211 Suppl 1: 53–61. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

