Induction of trained immunity by influenza vaccination - impact on COVID-19
- PMID: 34695164
- PMCID: PMC8568262
- DOI: 10.1371/journal.ppat.1009928
Induction of trained immunity by influenza vaccination - impact on COVID-19
Abstract
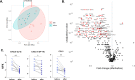
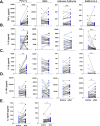
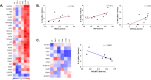
Non-specific protective effects of certain vaccines have been reported, and long-term boosting of innate immunity, termed trained immunity, has been proposed as one of the mechanisms mediating these effects. Several epidemiological studies suggested cross-protection between influenza vaccination and COVID-19. In a large academic Dutch hospital, we found that SARS-CoV-2 infection was less common among employees who had received a previous influenza vaccination: relative risk reductions of 37% and 49% were observed following influenza vaccination during the first and second COVID-19 waves, respectively. The quadrivalent inactivated influenza vaccine induced a trained immunity program that boosted innate immune responses against various viral stimuli and fine-tuned the anti-SARS-CoV-2 response, which may result in better protection against COVID-19. Influenza vaccination led to transcriptional reprogramming of monocytes and reduced systemic inflammation. These epidemiological and immunological data argue for potential benefits of influenza vaccination against COVID-19, and future randomized trials are warranted to test this possibility.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
-
- WHO. World health organisation, COVID-19 dashboard. https://covid19.who.int/. Accessed 18-03-2021, 2021.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

