Rapidly Declining SARS-CoV-2 Antibody Titers within 4 Months after BNT162b2 Vaccination
- PMID: 34696253
- PMCID: PMC8539465
- DOI: 10.3390/vaccines9101145
Rapidly Declining SARS-CoV-2 Antibody Titers within 4 Months after BNT162b2 Vaccination
Abstract
The efficacy and safety of the BNT162b2 vaccine are known, but antibodies are expected to decrease over time after vaccination. We collected blood samples from 104 fully vaccinated health care workers at 3 and 5 weeks after first vaccination and 4 months after second vaccination. Antibody titers and neutralizing antibodies were measured. In our study, both antibody titers and neutralizing antibodies increased significantly at 5 weeks after first vaccination but decreased rapidly at 4 months after second vaccination. Additionally, the results showed a significant decrease regardless of gender or age. Further studies are needed to help determine the interval of SARS-CoV-2 vaccinations.
Keywords: BNT162 vaccine; SARS-CoV-2; antibodies; neutralizing antibodies; vaccine immunogenicity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
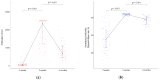
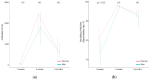
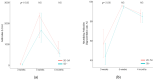
References
-
- WHO Coronavirus (COVID-19) Dashboard. [(accessed on 1 October 2021)]. Available online: https://covid19.who.int/
-
- Korea Centers for Disease and Prevention. [(accessed on 1 October 2021)]; Available online: http://www.kcdca.go.kr/
LinkOut - more resources
Full Text Sources
Miscellaneous

