Effects of Radix Polygalae on Cognitive Decline and Depression in Estradiol Depletion Mouse Model of Menopause
- PMID: 34698102
- PMCID: PMC8929121
- DOI: 10.3390/cimb43030118
Effects of Radix Polygalae on Cognitive Decline and Depression in Estradiol Depletion Mouse Model of Menopause
Abstract
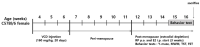
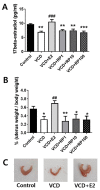
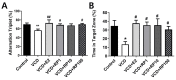
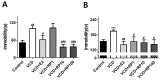
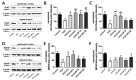
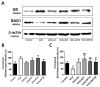
Postmenopausal syndrome refers to symptoms caused by the gradual decrease in female hormones after mid-40 years. As a target organ of estrogen, decrease in estrogen causes various changes in brain function such as a decrease in choline acetyltransferase and brain-derived neurotrophic factor; thus, postmenopausal women experience cognitive decline and more depressive symptoms than age-matched men. Radix Polygalae has been used for memory boosting and as a mood stabilizer and its components have shown neuroprotective, antidepressant, and stress relief properties. In a mouse model of estrogen depletion induced by 4-vinylcyclohexene diepoxide, Radix Polygalae was orally administered for 3 weeks. In these animals, cognitive and depression-related behaviors and molecular changes related to these behaviors were measured in the prefrontal cortex and hippocampus. Radix Polygalae improved working memory and contextual memory and despair-related behaviors in 4-vinylcyclohexene diepoxide-treated mice without increasing serum estradiol levels in this model. In relation to these behaviors, choline acetyltransferase and brain-derived neurotrophic factor in the prefrontal cortex and hippocampus and bcl-2-associated athanogene expression increased in the hippocampus. These results implicate the possible benefit of Radix Polygalae in use as a supplement of estrogen to prevent conditions such as postmenopausal depression and cognitive decline.
Keywords: VCD; cognition; depression; estrogen; radix polygalae.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

