Engineered antibody fusion proteins for targeted disease therapy
- PMID: 34706833
- PMCID: PMC8922469
- DOI: 10.1016/j.tips.2021.09.009
Engineered antibody fusion proteins for targeted disease therapy
Abstract
Since the FDA approval of the first therapeutic antibody 35 years ago, antibody-based products have gained prominence in the pharmaceutical market. Building on the early successes of monoclonal antibodies, more recent efforts have capitalized on the exquisite specificity and/or favorable pharmacokinetic properties of antibodies by developing fusion proteins that enable targeted delivery of therapeutic payloads which are otherwise ineffective when administered systemically. This review focuses on recent engineering and translational advances for therapeutics that genetically fuse antibodies to disease-relevant payloads, including cytokines, toxins, enzymes, neuroprotective agents, and soluble factor traps. With numerous antibody fusion proteins in the clinic and other innovative molecules poised to follow suit, these potent, multifunctional drug candidates promise to be a major player in the therapeutic development landscape for years to come.
Copyright © 2021 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no interests.
Figures

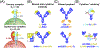

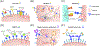
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

