Cardioversion in patients with newly diagnosed non-valvular atrial fibrillation: observational study using prospectively collected registry data
- PMID: 34706884
- PMCID: PMC8548918
- DOI: 10.1136/bmj-2021-066450
Cardioversion in patients with newly diagnosed non-valvular atrial fibrillation: observational study using prospectively collected registry data
Abstract
Objective: To investigate the clinical outcomes of patients who underwent cardioversion compared with those who did not have cardioverson in a large dataset of patients with recent onset non-valvular atrial fibrillation.
Design: Observational study using prospectively collected registry data (Global Anticoagulant Registry in the FIELD-AF-GARFIELD-AF).
Setting: 1317 participating sites in 35 countries.
Participants: 52 057 patients aged 18 years and older with newly diagnosed atrial fibrillation (up to six weeks' duration) and at least one investigator determined stroke risk factor.
Main outcome measures: Comparisons were made between patients who received cardioversion and those who had no cardioversion at baseline, and between patients who received direct current cardioversion and those who had pharmacological cardioversion. Overlap propensity weighting with Cox proportional hazards models was used to evaluate the effect of cardioversion on clinical endpoints (all cause mortality, non-haemorrhagic stroke or systemic embolism, and major bleeding), adjusting for baseline risk and patient selection.
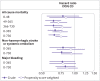
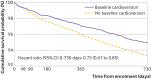
Results: 44 201 patients were included in the analysis comparing cardioversion and no cardioversion, and of these, 6595 (14.9%) underwent cardioversion at baseline. The propensity score weighted hazard ratio for all cause mortality in the cardioversion group was 0.74 (95% confidence interval 0.63 to 0.86) from baseline to one year follow-up and 0.77 (0.64 to 0.93) from one year to two year follow-up. Of the 6595 patients who had cardioversion at baseline, 299 had a follow-up cardioversion more than 48 days after enrolment. 7175 patients were assessed in the analysis comparing type of cardioversion: 2427 (33.8%) received pharmacological cardioversion and 4748 (66.2%) had direct current cardioversion. During one year follow-up, event rates (per 100 patient years) for all cause mortality in patients who received direct current and pharmacological cardioversion were 1.36 (1.13 to 1.64) and 1.70 (1.35 to 2.14), respectively.
Conclusion: In this large dataset of patients with recent onset non-valvular atrial fibrillation, a small proportion were treated with cardioversion. Direct current cardioversion was performed twice as often as pharmacological cardioversion, and there appeared to be no major difference in outcome events for these two cardioversion modalities. For the overall cardioversion group, after adjustments for confounders, a significantly lower risk of mortality was found in patients who received early cardioversion compared with those who did not receive early cardioversion.
Study registration: ClinicalTrials.gov NCT01090362.
© Author(s) (or their employer(s)) 2019. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: All authors have completed the ICMJE uniform disclosure form at www.icmje.org/disclosure-of-interest/ and declare: support from Kantor Charitable Foundation for the Kantor-Kakkar Global Centre for Thrombosis Science for the submitted work. TSH reports personal fees from AstraZeneca, Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Imedic, Novartis, MSD, Sanofi, and Pfizer. JYLH reports personal fees from Bayer, BMS/Pfizer, Boehringer Ingelheim, and Daiichi-Sankyo. PJ has served as a consultant or on an advisory board for Bayer, Boehringer Ingelheim, and Novartis. RC reports research grants from Boston Scientific, Medtronic, Abbott, Pfizer, Daiichi Sankyo, Biosense Webster, Boehringer Ingelheim, Johnson and Johnson, and personal fees from Boston Scientific, Medtronic, Biosense Webster, Abbott. DA reports personal fees from Bayer, Boehringer-Ingelheim, Bristol Meier Squibb, MSD and Pfizer, and grants to the institution from Medtronic and BMS. AJC has received institutional grant funding and personal fees from Bayer, Boehringer Ingelheim, Bristol Meier Squibb, Daiichi Sankyo, and Pfizer. AKK has received grants from Bayer AG, and Sanofi, personal fees from Bayer AG, Janssen, Pfizer, Sanofi, Verseon, and Anthos Therapeutics. All other authors have reported that they have no relationships relevant to the content of this paper to disclose.
Figures