Early-life inflammation primes a T helper 2 cell-fibroblast niche in skin
- PMID: 34707292
- PMCID: PMC8906225
- DOI: 10.1038/s41586-021-04044-7
Early-life inflammation primes a T helper 2 cell-fibroblast niche in skin
Abstract
Inflammation early in life can prime the local immune milieu of peripheral tissues, which can cause lasting changes in immunological tone that confer disease protection or susceptibility1. The cellular and molecular mechanisms that prompt changes in immune tone in many nonlymphoid tissues remain largely unknown. Here we find that time-limited neonatal inflammation induced by a transient reduction in neonatal regulatory T cells causes a dysregulation of subcutaneous tissue in mouse skin. This is accompanied by the selective accumulation of type 2 helper T (TH2) cells within a distinct microanatomical niche. TH2 cells are maintained into adulthood through interactions with a fibroblast population in skin fascia that we refer to as TH2-interacting fascial fibroblasts (TIFFs), which expand in response to TH2 cytokines to form subcutaneous fibrous bands. Activation of the TH2-TIFF niche due to neonatal inflammation primes the skin for altered reparative responses to wounding. Furthermore, we identify fibroblasts in healthy human skin that express the TIFF transcriptional signature and detect these cells at high levels in eosinophilic fasciitis, an orphan disease characterized by inflammation and fibrosis of the skin fascia. Taken together, these data define a previously unidentified TH2 cell niche in skin and functionally characterize a disease-associated fibroblast population. The results also suggest a mechanism of immunological priming whereby inflammation early in life creates networks between adaptive immune cells and stromal cells to establish an immunological set-point in tissues that is maintained throughout life.
© 2021. The Author(s), under exclusive licence to Springer Nature Limited.
Conflict of interest statement
Competing interests
Funding for scRNA-seq studies of healthy human skin was provided by LEO Pharmaceuticals. M.D.R. is a founder and consultant for TRex Bio., Sitryx Bio. and Mozart Therapeutics.
Figures


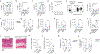







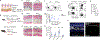
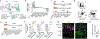

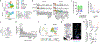
Comment in
-
Fibroblast niche sets TH2 skin tone in early life.Nat Rev Immunol. 2022 Jan;22(1):2-3. doi: 10.1038/s41577-021-00658-0. Nat Rev Immunol. 2022. PMID: 34782780 No abstract available.
References
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

