Messages that increase COVID-19 vaccine acceptance: Evidence from online experiments in six Latin American countries
- PMID: 34710168
- PMCID: PMC8553119
- DOI: 10.1371/journal.pone.0259059
Messages that increase COVID-19 vaccine acceptance: Evidence from online experiments in six Latin American countries
Erratum in
-
Correction: Messages that increase COVID-19 vaccine acceptance: Evidence from online experiments in six Latin American countries.PLoS One. 2025 Oct 29;20(10):e0335673. doi: 10.1371/journal.pone.0335673. eCollection 2025. PLoS One. 2025. PMID: 41160567 Free PMC article.
Abstract
As safe and effective vaccines become widely available, attaining herd immunity and limiting the spread of COVID-19 will depend on individuals choosing to vaccinate-and doing so quickly enough to outpace mutations. Using online surveys conducted across six Latin American countries in January 2021, we experimentally assess messages designed to counteract informational deficiencies and collective action problems that may drive hesitancy. We first find that basic vaccine information persuades around 8% of hesitant individuals to become willing to vaccinate, reduces intended wait to vaccinate by 0.4 months, and increases willingness to encourage others to vaccinate. Rather than facilitating free riding, learning, or social conformity, additional information about others' behavior increases vaccine acceptance when respondents expect herd immunity will be achieved. Finally, priming the social approval benefits of vaccinating also increases vaccine acceptance. These results suggest that providing information and shaping social expectations and incentives could both significantly increase vaccine uptake.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
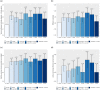
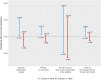
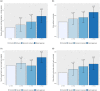
References
-
- Ipsos. COVID-19 vaccination intent has soared across the world. 2021. www.ipsos.com/en/covid-19-vaccination-intent-has-soared-across-world.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

