The Trp-rich Antimicrobial Amphiphiles With Intramolecular Aromatic Interactions for the Treatment of Bacterial Infection
- PMID: 34721331
- PMCID: PMC8548882
- DOI: 10.3389/fmicb.2021.733441
The Trp-rich Antimicrobial Amphiphiles With Intramolecular Aromatic Interactions for the Treatment of Bacterial Infection
Abstract
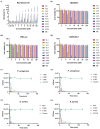
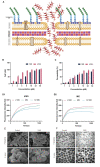
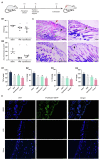
Antibiotic resistance is emerging as a hot issue with the abuse and overuse of antibiotics, and the shortage of effective antimicrobial agents against multidrug resistant bacteria creates a huge problem to treat the threatening nosocomial skin and soft tissue infection. Antimicrobial peptides (AMPs) exhibite enormous potential as one of the most promising candidates of antibiotic to fight against pathogenic infections because of its unique membrane penetration mechanism to kill pathogens, whereas the clinical application of AMPs still faces the challenges of production cost, stability, safety, and design strategy. Herein, a series of Trp-rich peptides was designed following the principle of paired Trp plated at the ith and ith+4 position on the backbone of peptides, based on the template (VKKX)4, where X represents W, A, or L, to study the effect of intramolecular aromatic interactions on the bioactivity of AMPs. Through comparing the antimicrobial performance, hemolysis, cytotoxicity, and stability, VW5 which is equipped with the characters of direct antimicrobial efficacy (GM=1.68μM) and physical destruction of bacterial membrane (SEM and electron microscopy) stood out from the engineering peptides. VW5 also performed well in mice models, which could significantly decrease the bacterial colony (VW5 vs infection group, 12.72±2.26 vs 5.52±2.01×109CFU/abscess), the area of dermo-necrosis (VW5 vs infection group, 0.74±0.29 vs 1.86±0.98mm2) and the inflammation cytokine levels at the abscess site without causing toxicity to the skin. Overall, this study provides a strategy and template to diminish the randomness in the exploration and design of novel peptides.
Keywords: antimicrobial activity; antimicrobial peptide; bacterial infection; intramolecular aromatic interaction; subcutaneous abscess.
Copyright © 2021 Wang, Li, Li, Li, Shang, Chou, Lyu and Shan.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

