Homologous Recombination Deficiency Assays in Epithelial Ovarian Cancer: Current Status and Future Direction
- PMID: 34722237
- PMCID: PMC8551835
- DOI: 10.3389/fonc.2021.675972
Homologous Recombination Deficiency Assays in Epithelial Ovarian Cancer: Current Status and Future Direction
Abstract
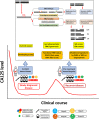
Epithelial ovarian cancer (EOC) patients are generally diagnosed at an advanced stage, usually relapse after initial treatments, which include debulking surgery and adjuvant platinum-based chemotherapy, and eventually have poor 5-year survival of less than 50%. In recent years, promising survival benefits from maintenance therapy with poly(ADP-ribose) polymerase (PARP) inhibitor (PARPi) has changed the management of EOC in newly diagnosed and recurrent disease. Identification of BRCA mutations and/or homologous recombination deficiency (HRD) is critical for selecting patients for PARPi treatment. However, the currently available HRD assays are not perfect predictors of the clinical response to PARPis in EOC patients. In this review, we introduce the concept of synthetic lethality, the rationale of using PARPi when HRD is present in tumor cells, the clinical trials of PARPi incorporating the HRD assays for EOC, the current HRD assays, and other HRD assays in development.
Keywords: PARP inhibitor; RAD51 foci formation; epithelial ovarian cancer; genomic scar; homologous recombination deficiency; mutational signatures.
Copyright © 2021 Chiang, Lin and Cheng.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Allemani C, Matsuda T, Di Carlo V, Harewood R, Matz M, Niksic M, et al. Global Surveillance of Trends in Cancer Survival 2000-14 (CONCORD-3): Analysis of Individual Records for 37 513 025 Patients Diagnosed With One of 18 Cancers From 322 Population-Based Registries in 71 Countries. Lancet (2018) 391(10125):1023–75. doi: 10.1016/S0140-6736(17)33326-3 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

