GRP78 Antibodies Are Associated With Blood-Brain Barrier Breakdown in Anti-Myelin Oligodendrocyte Glycoprotein Antibody-Associated Disorder
- PMID: 34725263
- PMCID: PMC8561843
- DOI: 10.1212/NXI.0000000000001038
GRP78 Antibodies Are Associated With Blood-Brain Barrier Breakdown in Anti-Myelin Oligodendrocyte Glycoprotein Antibody-Associated Disorder
Abstract
Background and objectives: To analyze (1) the effect of immunoglobulin G (IgG) from patients with anti-myelin oligodendrocyte glycoprotein antibody (MOG-Ab)-associated disorder on the blood-brain barrier (BBB) endothelial cells and (2) the positivity of glucose-regulated protein 78 (GRP78) antibodies in MOG-Ab-associated disorders.
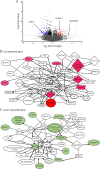
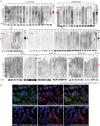
Methods: IgG was purified from sera with patients with MOG-Ab-associated disorder in the acute phase (acute MOG, n = 15), in the stable stage (stable MOG, n = 14), healthy controls (HCs, n = 9), and disease controls (DCs, n = 27). Human brain microvascular endothelial cells (BMECs) were incubated with IgG, and the number of nuclear NF-κB p65-positive cells in BMECs using high-content imaging system and the quantitative messenger RNA change in gene expression over the whole transcriptome using RNA-seq were analyzed. GRP78 antibodies from patient IgGs were detected by Western blotting.
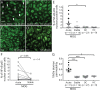
Results: IgG in the acute MOG group significantly induced the nuclear translocation of NF-κB and increased the vascular cell adhesion molecule 1/intercellular adhesion molecule 1 expression/permeability of 10-kDa dextran compared with that from the stable MOG and HC/DC groups. RNA-seq and pathway analysis revealed that NF-κB signaling and oxidative stress (NQO1) play key roles. The NQO1 and Nrf2 protein amounts were significantly decreased after exposure to IgG in the acute MOG group. The rate of GRP78 antibody positivity in the acute MOG group (10/15, 67% [95% confidence interval, 38%-88%]) was significantly higher than that in the stable MOG group (5/14, 36% [13%-65%]), multiple sclerosis group (4/29, 14% [4%-32%]), the DCs (3/27, 11% [2%-29%]), or HCs (0/9, 0%). Removal of GRP78 antibodies from MOG-IgG reduced the effect on NF-κB nuclear translocation and increased permeability.
Discussion: GRP78 antibodies may be associated with BBB dysfunction in MOG-Ab-associated disorder.
Copyright © 2021 The Author(s). Published by Wolters Kluwer Health, Inc. on behalf of the American Academy of Neurology.
Figures



References
-
- Cobo-Calvo A, Vukusic S, Marignier R. Clinical spectrum of central nervous system myelin oligodendrocyte glycoprotein autoimmunity in adults. Curr Opin Neurol. 2019;32(3):459-466. - PubMed
-
- Reindl M, Jarius S, Rostasy K, Berger T. Myelin oligodendrocyte glycoprotein antibodies: how clinically useful are they? Curr Opin Neurol. 2017;30(3):295-301. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
