Activation of parabrachial nucleus - ventral tegmental area pathway underlies the comorbid depression in chronic neuropathic pain in mice
- PMID: 34731609
- PMCID: PMC8578703
- DOI: 10.1016/j.celrep.2021.109936
Activation of parabrachial nucleus - ventral tegmental area pathway underlies the comorbid depression in chronic neuropathic pain in mice
Abstract
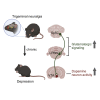
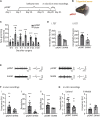
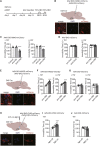
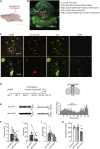
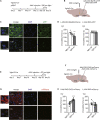
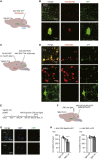
Depression symptoms are often found in patients suffering from chronic pain, a phenomenon that is yet to be understood mechanistically. Here, we systematically investigate the cellular mechanisms and circuits underlying the chronic-pain-induced depression behavior. We show that the development of chronic pain is accompanied by depressive-like behaviors in a mouse model of trigeminal neuralgia. In parallel, we observe increased activity of the dopaminergic (DA) neuron in the midbrain ventral tegmental area (VTA), and inhibition of this elevated VTA DA neuron activity reverses the behavioral manifestations of depression. Further studies establish a pathway of glutamatergic projections from the spinal trigeminal subnucleus caudalis (Sp5C) to the lateral parabrachial nucleus (LPBN) and then to the VTA. These glutamatergic projections form a direct circuit that controls the development of the depression-like behavior under the state of the chronic neuropathic pain.
Copyright © 2021 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures

References
-
- Arnow B.A., Hunkeler E.M., Blasey C.M., Lee J., Constantino M.J., Fireman B., Kraemer H.C., Dea R., Robinson R., Hayward C. Comorbid depression, chronic pain, and disability in primary care. Psychosom. Med. 2006;68:262–268. - PubMed
-
- Bair M.J., Robinson R.L., Katon W., Kroenke K. Depression and pain comorbidity: a literature review. Arch. Intern. Med. 2003;163:2433–2445. - PubMed
-
- Bernard J.F., Bester H., Besson J.M. Involvement of the spino-parabrachio -amygdaloid and -hypothalamic pathways in the autonomic and affective emotional aspects of pain. Prog. Brain Res. 1996;107:243–255. - PubMed
-
- Berridge K.C. Comparative Fine Structure of Action: Rules of Form and Sequence in the Grooming Patterns of Six Rodent Species. Behaviour. 1990;113:21–56.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

