A Severe Case of Idiosyncratic Hepatotoxicity with Unfractionated Heparin
- PMID: 34732938
- PMCID: PMC8559046
- DOI: 10.1177/0018578720965435
A Severe Case of Idiosyncratic Hepatotoxicity with Unfractionated Heparin
Abstract
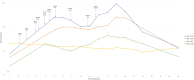
The authors describe a case of clinically apparent idiosyncratic hepatotoxicity in association with unfractionated heparin (UFH). A 52-year-old woman with increasingly symptomatic rheumatic mitral valvular disease and severe pulmonary hypertension underwent elective minimally-invasive bioprosthetic mitral valve replacement. The patient received 42 000 units of UFH intraoperatively 10 days after receiving 3100 units during a left heart catheterization. Standard prophylactic doses of unfractionated heparin were started on POD 2 for prevention of venous thromboembolism. On the evening of postoperative day (POD) 3, the patient was lethargic, encephalopathic, and hypoglycemic with an acute liver injury and hyperlactatemia. Similar events occurred on POD 7 after clinical improvement from the initial injury and an unintentional rechallenge with UFH. Heparins are usually not suspected of idiosyncratic hepatotoxicity due to their widespread utilization and reports of milder episodes of hepatotoxicity. This case highlights the need to consider UFH in the differential of drug-induced liver injury, including severe cases.
Keywords: adverse drug reactions; anticoagulants; cardiovascular; gastrointestinal disorders.
© The Author(s) 2020.
Conflict of interest statement
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures
References
-
- National Institutes of Health. Heparins. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. Bethesda, MD: National Institutes of Health; 2012. https://www.ncbi.nlm.nih.gov/books/NBK548770/. Updated November 13, 2017.Accessed April 23, 2020.
-
- National Institutes of Health. Low Molecular Weight Heparins. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. Bethesda, MD: National Institutes of Health; 2012. https://www.ncbi.nlm.nih.gov/books/NBK548009/.Updated November 13, 2017. Accessed April 23, 2020.
LinkOut - more resources
Full Text Sources

