Building an institutional K awardee program at UC Davis through utilization of CTSA resources
- PMID: 34733547
- PMCID: PMC8532183
- DOI: 10.1017/cts.2021.839
Building an institutional K awardee program at UC Davis through utilization of CTSA resources
Abstract
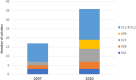
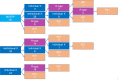
NIH offers multiple mentored career development award mechanisms. By building on the UC Davis Clinical and Translational Science Center (CTSC) from its initial NIH funding in 2006, we created an institution-wide K scholar resource. We investigated subsequent NIH funding for K scholars and to what extent CTSC research resources were used. Using NIH RePORTER, we created a database of UC Davis investigators who obtained K01, K08, K23, K25, or K99, as well as institutional KL2 or K12 awards and tracked CTSC research resource use and subsequent funding success. Overall, 94 scholars completed K training between 2007 and 2020, of which 70 participated in one of four institutional, NIH-funded K programs. An additional 103 scholars completed a mentored clinical research training program. Of 94 K awardees, 61 (65%) later achieved NIH funding, with the majority receiving a subsequent individual K award. A higher proportion (73%) of funded scholars used CTSC resources compared to unfunded (48%). Biostatistics and Biomedical Informatics were most commonly used and 55% of scholars used one or more CTSC resource. We conclude that institutional commitment to create a K scholar platform and use of CTSC research resources is associated with high NIH funding rates for early career investigators.
Keywords: K awards; NIH funding; career development; clinical and translational science awards; mentored.
© The Author(s) 2021.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Figures
References
-
- National Institutes of Health Advisory Committee to the Director. Biomedical Workforce Working Group Report [Internet]. (https://acd.od.nih.gov/documents/reports/Biomedical_research_wgreport.pdf)
-
- Zerhouni EA. Clinical research at a crossroads: the NIH roadmap. Journal of Investigative Medicine 2006; 54(4): 171–173. - PubMed
-
- Reis SE, Berglund L, Bernard GR, Califf RM, FitzGerald GA, Johnson PC. A roadmap for re-engineering the national clinical and translational research enterprise: the national clinical and translational science award (CTSA) consortium’s strategic plan. Academic Medicine 2010; 85(3): 463–469. - PMC - PubMed
-
- Meyers FJ, Begg MD, Fleming M, Merchant C. Strengthening the career development of clinical translational scientist trainees: a consensus statement of the clinical translational science award (CTSA) research education and career development committees. Clinical and Translational Science 2012; 5(2): 123–127. - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous
