Engineering Strategies for Immunomodulatory Cytokine Therapies - Challenges and Clinical Progress
- PMID: 34734110
- PMCID: PMC8562465
- DOI: 10.1002/adtp.202100035
Engineering Strategies for Immunomodulatory Cytokine Therapies - Challenges and Clinical Progress
Abstract
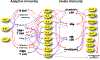
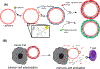
Cytokines are immunoregulatory proteins involved in many pathological states with promising potential as therapeutic agents. A diverse array of cytokines have been studied in preclinical disease models since the 1950s, some of which became successful biopharmaceutical products with the advancement of recombinant protein technology in the 1980s. However, following these early approvals, clinical translation of these natural immune signaling molecules has been limited due to their pleiotropic action in many cell types, and the fact that they have evolved to act primarily locally in tissues. These characteristics, combined with poor pharmacokinetics, have hindered the delivery of cytokines via systemic administration routes due to dose-limiting toxicities. However, given their clinical potential and recent clinical successes in cancer immunotherapy, cytokines continue to be extensively pursued in preclinical and clinical studies, and a range of molecular and formulation engineering strategies are being applied to reduce treatment toxicity while maintaining or enhancing therapeutic efficacy. This review provides a brief background on the characteristics of cytokines and their history as clinical therapeutics, followed by a deeper discussion on the engineering strategies developed for cytokine therapies with a focus on the translational relevance of these approaches.
Keywords: clinical trials; cytokines; drug delivery; immunotherapies; therapeutics.
Figures





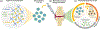


References
-
- Cohen S, Bigazzi PE, Yoshida T, Similarities of T cell function in cell-mediated immunity and antibody production, Vol. 12, Academic Press, 1974, pp. 150–159. - PubMed
-
- Kronfol Z, Cytokines and mental health, Kluwer Academic Pub, 2003.
-
- Caligiuri MA, Lotze MT, Cytokines in the genesis and treatment of cancer, Humana Press, 2007.
-
- Wang C, Ye Y, Hu Q, Bellotti A, Gu Z, Tailoring Biomaterials for Cancer Immunotherapy: Emerging Trends and Future Outlook, Vol. 29, Wiley-VCH Verlag, 2017. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
