Non-Randomized Trial of Dornase Alfa for Acute Respiratory Distress Syndrome Secondary to Covid-19
- PMID: 34745093
- PMCID: PMC8564175
- DOI: 10.3389/fimmu.2021.714833
Non-Randomized Trial of Dornase Alfa for Acute Respiratory Distress Syndrome Secondary to Covid-19
Abstract
Background: The most severe cases of Coronavirus-Disease-2019 (COVID-19) develop into Acute Respiratory Distress Syndrome (ARDS). It has been proposed that oxygenation may be inhibited by extracellular deoxyribonucleic acid (DNA) in the form of neutrophil extracellular traps (NETs). Dornase alfa (Pulmozyme, Genentech) is recombinant human deoxyribonuclease I that acts as a mucolytic by cleaving and degrading extracellular DNA. We performed a pilot study to evaluate the effects of dornase alfa in patients with ARDS secondary to COVID-19.
Methods: We performed a pilot, non-randomized, case-controlled clinical trial of inhaled dornase for patients who developed ARDS secondary to COVID-19 pneumonia.
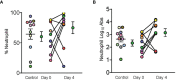
Results: Improvement in arterial oxygen saturation to inhaled fraction of oxygen ratio (PaO2/FiO2) was noted in the treatment group compared to control at day 2 (95% CI, 2.96 to 95.66, P-value = 0.038), as well as in static lung compliance at days 3 through 5 (95% CI, 4.8 to 19.1 mL/cmH2O, 2.7 to 16.5 mL/cmH2O, and 5.3 to 19.2 mL/cmH2O, respectively). These effects were not sustained at 14 days. A reduction in bronchoalveolar lavage fluid (BALF) myeloperoxidase-DNA (DNA : MPO) complexes (95% CI, -14.7 to -1.32, P-value = 0.01) was observed after therapy with dornase alfa.
Conclusion: Treatment with dornase alfa was associated with improved oxygenation and decreased DNA : MPO complexes in BALF. The positive effects, however, were limited to the time of drug delivery. These data suggest that degradation of extracellular DNA associated with NETs or other structures by inhaled dornase alfa can be beneficial. We propose a more extensive clinical trial is warranted.
Clinical trial registration: ClinicalTrials.gov, Identifier: NCT04402970.
Keywords: ARDS; COVID - 19; pneumonia; respiratory failure; viral.
Copyright © 2021 Holliday, Earhart, Alnijoumi, Krvavac, Allen and Schrum.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

