The right time for senescence
- PMID: 34756162
- PMCID: PMC8580479
- DOI: 10.7554/eLife.72449
The right time for senescence
Abstract
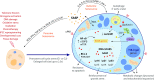
Cellular senescence is a highly complex and programmed cellular state with diverse and, at times, conflicting physiological and pathological roles across the lifespan of an organism. Initially considered a cell culture artifact, senescence evolved from an age-related circumstance to an intricate cellular defense mechanism in response to stress, implicated in a wide spectrum of biological processes like tissue remodelling, injury and cancer. The development of new tools to study senescence in vivo paved the way to uncover its functional roles in various frameworks, which are sometimes hard to reconcile. Here, we review the functional impact of senescent cells on different organismal contexts. We provide updated insights on the role of senescent cells in tissue repair and regeneration, in which they essentially modulate the levels of fibrosis and inflammation, discussing how "time" seems to be the key maestro of their effects. Finally, we overview the current clinical research landscape to target senescent cells and contemplate its repercussions on this fast-evolving field.
Keywords: cell biology; pathophysiology; senescence; senotherapies; time; tissue remodelling.
© 2021, Paramos-de-Carvalho et al.
Conflict of interest statement
DP, AJ, LS No competing interests declared
Figures

References
-
- Acosta JC, Banito A, Wuestefeld T, Georgilis A, Janich P, Morton JP, Athineos D, Kang T-W, Lasitschka F, Andrulis M, Pascual G, Morris KJ, Khan S, Jin H, Dharmalingam G, Snijders AP, Carroll T, Capper D, Pritchard C, Inman GJ, Longerich T, Sansom OJ, Benitah SA, Zender L, Gil J. A complex secretory program orchestrated by the inflammasome controls paracrine senescence. Nature Cell Biology. 2013;15:978–990. doi: 10.1038/ncb2784. - DOI - PMC - PubMed
-
- Amor C, Feucht J, Leibold J, Ho YJ, Zhu C, Alonso-Curbelo D, Mansilla-Soto J, Boyer JA, Li X, Giavridis T, Kulick A, Houlihan S, Peerschke E, Friedman SL, Ponomarev V, Piersigilli A, Sadelain M, Lowe SW. Senolytic CAR T cells reverse senescence-associated pathologies. Nature. 2020;583:127–132. doi: 10.1038/s41586-020-2403-9. - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

