Prognostic Value of Resistance Proteins in Plasma Cells from Multiple Myeloma Patients Treated with Bortezomib-Based Regimens
- PMID: 34768548
- PMCID: PMC8584776
- DOI: 10.3390/jcm10215028
Prognostic Value of Resistance Proteins in Plasma Cells from Multiple Myeloma Patients Treated with Bortezomib-Based Regimens
Abstract
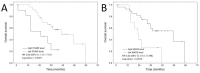
While multiple myeloma (MM) treatment with proteasome inhibitors and other agents yields encouraging results, primary and secondary resistance remains an emerging problem. An important factor in such treatment resistance is the overexpression of several proteins. The present study comprehensively evaluates the expression of POMP, PSMB5, NRF2, XBP1, cMAF and MAFb proteins in plasma cells isolated from the bone marrow of 39 MM patients treated with bortezomib-based regimens using an enzyme-linked immunosorbent assay (ELISA). The proteins were selected on the basis of previous laboratory and clinical studies in bortezomib-treated MM patients. It was found that the expression of the investigated proteins did not significantly differ between bortezomib-sensitive and bortezomib-refractory patients. However, the expression of some proteins correlated with overall survival (OS); this was significantly shorter in patients with higher POMP expression (HR 2.8, 95% CI: 1.1-7.0, p = 0.0277) and longer in those with higher MAFB expression (HR 0.32, 95% CI: 0.13-0.80, p = 0.0147). Our results indicate that a high expression of POMP and MAFB in MM plasma cells may serve as predictors of OS in MM patients treated with bortezomib-based regimens. However, further studies are needed to determine the role of these factors in effective strategies for improving anti-myeloma therapy.
Keywords: MAFb; OS; PFS; POMP; bortezomib; cMAF; multiple myeloma; prognosis; survival.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
References
-
- Robak P., Robak T. Novel drugs for multiple myeloma. Top. Anti-Cancer Res. 2019;8:1–43.
Grants and funding
LinkOut - more resources
Full Text Sources

