Preparation and Characterization of Water-Insoluble Gardenia Blue Pigment
- PMID: 34772116
- PMCID: PMC8585408
- DOI: 10.3390/ma14216594
Preparation and Characterization of Water-Insoluble Gardenia Blue Pigment
Abstract
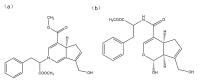
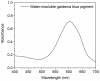
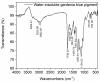
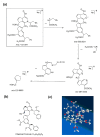
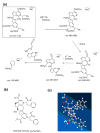
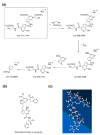
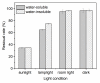
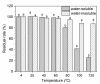
Based on molecular simulations, the synthetic route of water-insoluble gardenia blue pigment was prepared by the reaction of genipin and L-Phenylalanine methyl ester hydrochloride. A highly purified pigment was obtained after extraction by chloroform and purification by silica gel column chromatography, and the value of color is up to 288. A study on the structural characteristics of the pigment was implemented with a scanning electron microscope, ultraviolet-visible spectrophotometer, Fourier transform infrared spectrometer, X-ray photoelectron spectrometer, and quatropde-time of flight mass spectrometer. The results showed that the surface of the pigment was largely smooth and spherical; The λmax was 607 nm, and the main functional groups include O-C=O, C=O, C-N, C=C, OH, and benzene ring; We detrained six different molecular weight and chemical structures of pigments and speculated the particular structures and formation mechanisms of three kinds of pigment, whose molecular weights are 690.1156, 720.1226, and 708.1246 Da, respectively. The pigment was only able to be dissolved in ethanol, methanol, acetone, ethyl acetate, and other strong polar organic solvents, but was not able to be dissolved in water, ethyl ether, petroleum ether, and other weak polar organic solvents. In terms of light and thermal stabilities, water-insoluble gardenia blue pigment is significantly better than water-soluble gardenia blue pigment (p < 0.05). When it is under direct light for 7 days or incubated at 80-120 °C for 24 h, the pigment residual rates were 74.90, 95.26, 88.27, and 87.72%, respectively.
Keywords: gardenia blue pigment; solubility study; stability study; structure study; water-insoluble.
Conflict of interest statement
There is no conflict of interests regarding the publication of this paper.
Figures


Similar articles
-
Hydrophobic blue pigment formation from phosphatidylgenipin.J Nutr Sci Vitaminol (Tokyo). 1994 Oct;40(5):505-9. doi: 10.3177/jnsv.40.505. J Nutr Sci Vitaminol (Tokyo). 1994. PMID: 7891211
-
Purification and characterization of indochrome type blue pigment produced by Pseudarthrobacter sp. 34LCH1 isolated from Atacama desert.J Ind Microbiol Biotechnol. 2019 Jan;46(1):101-111. doi: 10.1007/s10295-018-2088-3. Epub 2018 Oct 30. J Ind Microbiol Biotechnol. 2019. PMID: 30377866
-
Preparation of a genipin blue from egg protein and genipin.Nat Prod Res. 2012;26(8):765-9. doi: 10.1080/14786419.2010.547859. Epub 2011 Sep 6. Nat Prod Res. 2012. PMID: 21895461
-
Gardenia jasminoides Ellis: Ethnopharmacology, phytochemistry, and pharmacological and industrial applications of an important traditional Chinese medicine.J Ethnopharmacol. 2020 Jul 15;257:112829. doi: 10.1016/j.jep.2020.112829. Epub 2020 Apr 18. J Ethnopharmacol. 2020. PMID: 32311486 Review.
-
FTIR study of primate color visual pigments.Biophysics (Nagoya-shi). 2015 Mar 4;11:61-6. doi: 10.2142/biophysics.11.61. eCollection 2015. Biophysics (Nagoya-shi). 2015. PMID: 27493516 Free PMC article. Review.
References
-
- Jespersen L., Stromdahl L.D., Olsen K., Skibsted L.H. Heat and light stability of three natural blue colorants for use in confectionery and beverages. Eur. Food Res. Technol. 2005;220:261–266. doi: 10.1007/s00217-004-1062-7. - DOI
-
- Wu S., Wu Q., Xu L., Xu L.J. Progress in the development and application of natural edible pigment. Shandong Food Ferment. 2015;179:35–38.
-
- Cho Y.J., Kim S.Y., Kim J., Choe E.K., Kim S.I., Shin H.J. One-step enzymatic synthesis of blue pigments from geniposide for fabric dyeing. Biotechnol. Bioprocess Eng. 2006;11:230–234. doi: 10.1007/BF02932035. - DOI
-
- Kim S.J., Jang H.G. Characterization and stability of Gardenia jasminoides biotransformed pigment produced in jar fermentor. J. Korean Soc. Food Sci. Nutr. 2005;34:880–884.
LinkOut - more resources
Full Text Sources

