The Use of Induced Pluripotent Stem Cells to Study the Effects of Adenosine Deaminase Deficiency on Human Neutrophil Development
- PMID: 34777360
- PMCID: PMC8582638
- DOI: 10.3389/fimmu.2021.748519
The Use of Induced Pluripotent Stem Cells to Study the Effects of Adenosine Deaminase Deficiency on Human Neutrophil Development
Abstract
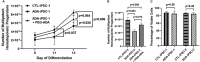
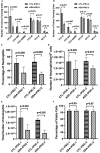
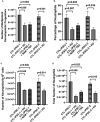
Inherited defects that abrogate the function of the adenosine deaminase (ADA) enzyme and consequently lead to the accumulation of toxic purine metabolites cause profound lymphopenia and severe combined immune deficiency. Additionally, neutropenia and impaired neutrophil function have been reported among ADA-deficient patients. However, due to the rarity of the disorder, the neutrophil developmental abnormalities and the mechanisms contributing to them have not been characterized. Induced pluripotent stem cells (iPSC) generated from two unrelated ADA-deficient patients and from healthy controls were differentiated through embryoid bodies into neutrophils. ADA deficiency led to a significant reduction in the number of all early multipotent hematopoietic progenitors. At later stages of differentiation, ADA deficiency impeded the formation of granulocyte colonies in methylcellulose cultures, leading to a significant decrease in the number of neutrophils generated from ADA-deficient iPSCs. The viability and apoptosis of ADA-deficient neutrophils isolated from methylcellulose cultures were unaffected, suggesting that the abnormal purine homeostasis in this condition interferes with differentiation or proliferation. Additionally, there was a significant increase in the percentage of hyperlobular ADA-deficient neutrophils, and these neutrophils demonstrated significantly reduced ability to phagocytize fluorescent microspheres. Supplementing iPSCs and methylcellulose cultures with exogenous ADA, which can correct adenosine metabolism, reversed all abnormalities, cementing the critical role of ADA in neutrophil development. Moreover, chemical inhibition of the ribonucleotide reductase (RNR) enzyme, using hydroxyurea or a combination of nicotinamide and trichostatin A in iPSCs from healthy controls, led to abnormal neutrophil differentiation similar to that observed in ADA deficiency, implicating RNR inhibition as a potential mechanism for the neutrophil abnormalities. In conclusion, the findings presented here demonstrate the important role of ADA in the development and function of neutrophils while clarifying the mechanisms responsible for the neutrophil abnormalities in ADA-deficient patients.
Keywords: adenosine deaminase (ADA) deficiency; iPSC (induced pluripotent stem cell); multipotent; neutropenia; ribonucleotide reductase (RNR) inhibitors.
Copyright © 2021 Tsui, Min, Ng, Dobbs, Notarangelo, Dror and Grunebaum.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Alterations in the adenosine metabolism and CD39/CD73 adenosinergic machinery cause loss of Treg cell function and autoimmunity in ADA-deficient SCID.Blood. 2012 Feb 9;119(6):1428-39. doi: 10.1182/blood-2011-07-366781. Epub 2011 Dec 19. Blood. 2012. PMID: 22184407 Free PMC article. Clinical Trial.
-
ADA Deficiency: Evaluation of the Clinical and Laboratory Features and the Outcome.J Clin Immunol. 2018 May;38(4):484-493. doi: 10.1007/s10875-018-0496-9. Epub 2018 May 9. J Clin Immunol. 2018. PMID: 29744787
-
Early Enzyme Replacement Therapy Improves Hearing and Immune Defects in Adenosine Deaminase Deficient-Mice.Front Immunol. 2019 Mar 13;10:416. doi: 10.3389/fimmu.2019.00416. eCollection 2019. Front Immunol. 2019. PMID: 30918508 Free PMC article.
-
Recent advances in understanding and managing adenosine deaminase and purine nucleoside phosphorylase deficiencies.Curr Opin Allergy Clin Immunol. 2013 Dec;13(6):630-8. doi: 10.1097/ACI.0000000000000006. Curr Opin Allergy Clin Immunol. 2013. PMID: 24113229 Review.
-
Adenosine Deaminase (ADA)-Deficient Severe Combined Immune Deficiency (SCID): Molecular Pathogenesis and Clinical Manifestations.J Clin Immunol. 2017 Oct;37(7):626-637. doi: 10.1007/s10875-017-0433-3. Epub 2017 Aug 25. J Clin Immunol. 2017. PMID: 28842866 Review.
Cited by
-
Neutrophil heterogeneity and aging: implications for COVID-19 and wound healing.Front Immunol. 2023 Nov 28;14:1201651. doi: 10.3389/fimmu.2023.1201651. eCollection 2023. Front Immunol. 2023. PMID: 38090596 Free PMC article. Review.
-
Purine nucleoside phosphorylase deficiency induces p53-mediated intrinsic apoptosis in human induced pluripotent stem cell-derived neurons.Sci Rep. 2022 May 31;12(1):9084. doi: 10.1038/s41598-022-10935-0. Sci Rep. 2022. PMID: 35641516 Free PMC article.
-
Roles of mitochondria in neutrophils.Front Immunol. 2022 Aug 19;13:934444. doi: 10.3389/fimmu.2022.934444. eCollection 2022. Front Immunol. 2022. PMID: 36081497 Free PMC article. Review.
-
Deficiency of Human Adenosine Deaminase Type 2 - A Diagnostic Conundrum for the Hematologist.Front Immunol. 2022 May 3;13:869570. doi: 10.3389/fimmu.2022.869570. eCollection 2022. Front Immunol. 2022. PMID: 35592317 Free PMC article. Review.
-
In vitro systems to study inborn errors of immunity using human induced pluripotent stem cells.Front Immunol. 2022 Nov 17;13:1024935. doi: 10.3389/fimmu.2022.1024935. eCollection 2022. Front Immunol. 2022. PMID: 36466870 Free PMC article. Review.
References
-
- Sauer AV, Brigida I, Carriglio N, Hernandez RJ, Scaramuzza S, Clavenna D, et al. . Alterations in the Adenosine Metabolism and CD39/CD73 Adenosinergic Machinery Cause Loss of Treg Cell Function and Autoimmunity in ADA-Deficient SCID. Blood (2012) 119(6):1428–39. doi: 10.1182/blood-2011-07-366781 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

