Neuromuscular Development and Disease: Learning From in vitro and in vivo Models
- PMID: 34778273
- PMCID: PMC8579029
- DOI: 10.3389/fcell.2021.764732
Neuromuscular Development and Disease: Learning From in vitro and in vivo Models
Abstract
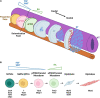
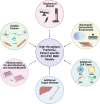
The neuromuscular junction (NMJ) is a specialized cholinergic synaptic interface between a motor neuron and a skeletal muscle fiber that translates presynaptic electrical impulses into motor function. NMJ formation and maintenance require tightly regulated signaling and cellular communication among motor neurons, myogenic cells, and Schwann cells. Neuromuscular diseases (NMDs) can result in loss of NMJ function and motor input leading to paralysis or even death. Although small animal models have been instrumental in advancing our understanding of the NMJ structure and function, the complexities of studying this multi-tissue system in vivo and poor clinical outcomes of candidate therapies developed in small animal models has driven the need for in vitro models of functional human NMJ to complement animal studies. In this review, we discuss prevailing models of NMDs and highlight the current progress and ongoing challenges in developing human iPSC-derived (hiPSC) 3D cell culture models of functional NMJs. We first review in vivo development of motor neurons, skeletal muscle, Schwann cells, and the NMJ alongside current methods for directing the differentiation of relevant cell types from hiPSCs. We further compare the efficacy of modeling NMDs in animals and human cell culture systems in the context of five NMDs: amyotrophic lateral sclerosis, myasthenia gravis, Duchenne muscular dystrophy, myotonic dystrophy, and Pompe disease. Finally, we discuss further work necessary for hiPSC-derived NMJ models to function as effective personalized NMD platforms.
Keywords: disease modeling; drug development; human skeletal muscle; induced pluripotent stem cells; muscular dystrophy; neuromuscular junction; organ on a chip; tissue engineering.
Copyright © 2021 Fralish, Lotz, Chavez, Khodabukus and Bursac.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

