Epsin 3 potentiates the NF‑κB signaling pathway to regulate apoptosis in breast cancer
- PMID: 34779498
- PMCID: PMC8600415
- DOI: 10.3892/mmr.2021.12531
Epsin 3 potentiates the NF‑κB signaling pathway to regulate apoptosis in breast cancer
Abstract
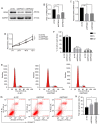
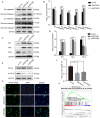
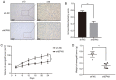
Endocrine drug resistance is common in some patients with estrogen receptor (ER)‑positive breast cancer, so it is necessary to identify potential therapeutic targets. The aim of the present study was to investigate the regulatory effect and mechanism of epsin 3 (EPN3) expression level changes on the proliferation and apoptosis of ER‑positive breast cancer. Online GEPIA was used to analyze the expression level of EPN3 in breast cancer. The online Kaplan‑Meier plotter tool was used to analyze the relationship between EPN3 expression and the prognosis of patients with breast cancer. Reverse transcription‑quantitative PCR, immunohistochemistry and western blotting were performed to detect the mRNA and protein expression levels of EPN3 in breast cancer tissues and cells. A lentiviral infection system was used to knockdown the expression of EPN3 in breast cancer cell lines. Cell Counting Kit‑8 and flow cytometry assays were conducted to detect the effect of EPN3 knockdown on breast cancer cell proliferation and apoptosis. Western blotting was used to detect the regulation of EPN3 expression on NF‑κB, and immunofluorescence was performed to detect the effect of EPN3 expression on NF‑κB nuclear translocation. The results demonstrated that the expression level of EPN3 in breast cancer tissues was higher compared with that in adjacent tissues (P<0.05). The expression level of EPN3 in the ER‑positive breast cancer cell line, MCF7, was higher compared with that in the other cell lines (MCF10A, ZR75‑1, MDA‑MB‑231, BT549 and SK‑BR‑3). After knocking down the expression of EPN3 in MCF7 cells, the proliferative ability of the cells was decreased, and the apoptosis rate was increased (P<0.05). After EPN3 knockdown in MCF7 cells, the phosphorylation of NF‑κB was decreased (P<0.05), and the nuclear translocation signal was weakened. Thus, it was suggested that EPN3 promoted cell proliferation and inhibited cell apoptosis by regulating the NF‑κB signaling pathway in ER‑positive breast cancer.
Keywords: NF‑κB; apoptosis; epsin 3; estrogen receptor‑positive breast cancer.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


Similar articles
-
Overexpression of Epsin 3 enhances migration and invasion of glioma cells by inducing epithelial‑mesenchymal transition.Oncol Rep. 2018 Nov;40(5):3049-3059. doi: 10.3892/or.2018.6691. Epub 2018 Sep 10. Oncol Rep. 2018. PMID: 30226603
-
EPN3 plays oncogenic role in non-small cell lung cancer by activating the JAK1/2-STAT3 pathway.Environ Toxicol. 2023 Aug;38(8):1968-1979. doi: 10.1002/tox.23822. Epub 2023 Apr 26. Environ Toxicol. 2023. PMID: 37186036
-
IGJ suppresses breast cancer growth and metastasis by inhibiting EMT via the NF‑κB signaling pathway.Int J Oncol. 2023 Sep;63(3):105. doi: 10.3892/ijo.2023.5553. Epub 2023 Aug 4. Int J Oncol. 2023. PMID: 37539706 Free PMC article.
-
miR‑302d‑3p regulates the viability, migration and apoptosis of breast cancer cells through regulating the TMBIM6‑mediated ERK signaling pathway.Mol Med Rep. 2021 Dec;24(6):853. doi: 10.3892/mmr.2021.12493. Epub 2021 Oct 15. Mol Med Rep. 2021. PMID: 34651659 Free PMC article.
-
NFκB affects estrogen receptor expression and activity in breast cancer through multiple mechanisms.Mol Cell Endocrinol. 2015 Dec 15;418 Pt 3(0 3):235-9. doi: 10.1016/j.mce.2014.09.013. Epub 2014 Oct 18. Mol Cell Endocrinol. 2015. PMID: 25450861 Free PMC article. Review.
Cited by
-
Discovering Breast Cancer Biomarkers Candidates through mRNA Expression Analysis Based on The Cancer Genome Atlas Database.J Pers Med. 2022 Oct 21;12(10):1753. doi: 10.3390/jpm12101753. J Pers Med. 2022. PMID: 36294892 Free PMC article.
-
SB218078 inhibits angiogenesis and epithelial-mesenchymal transition in breast cancer.Front Pharmacol. 2025 Mar 14;16:1552707. doi: 10.3389/fphar.2025.1552707. eCollection 2025. Front Pharmacol. 2025. PMID: 40160462 Free PMC article.
-
Epsin3 promotes non-small cell lung cancer progression via modulating EGFR stability.Cell Biosci. 2025 Feb 5;15(1):14. doi: 10.1186/s13578-025-01358-1. Cell Biosci. 2025. PMID: 39910656 Free PMC article.
References
-
- Goldhirsch A, Wood WC, Coates AS, Gelber RD, Thürlimann B, Senn HJ, Members P. Strategies for subtypes-dealing with the diversity of breast cancer: Highlights of the St. Gallen international expert consensus on the primary therapy of early breast cancer 2011. Ann Oncol. 2011;22:1736–1747. doi: 10.1093/annonc/mdr304. - DOI - PMC - PubMed
-
- Sotiriou C, Neo SY, McShane LM, Korn EL, Long PM, Jazaeri A, Martiat P, Fox SB, Harris AL, Liu ET. Breast cancer classification and prognosis based on gene expression profiles from a population-based study. Proc Natl Acad Sci USA. 2003;100:10393–10398. doi: 10.1073/pnas.1732912100. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

