Device-Based Enrichment of Knee Joint Synovial Cells to Drive MSC Chondrogenesis Without Prior Culture Expansion In Vitro: A Step Closer to 1-Stage Orthopaedic Procedures
- PMID: 34779670
- PMCID: PMC8739599
- DOI: 10.1177/03635465211055164
Device-Based Enrichment of Knee Joint Synovial Cells to Drive MSC Chondrogenesis Without Prior Culture Expansion In Vitro: A Step Closer to 1-Stage Orthopaedic Procedures
Abstract
Background: Synovial fluid (SF) mesenchymal stem cells (MSCs) are derived from the synovial membrane and have cartilage repair potential. Their current use in clinical practice is largely exploratory. As their numbers tend to be small, therapeutic procedures using MSCs typically require culture expansion. Previous reports indicate that the stem cell-mobilizing device (STEM device) intraoperatively increases SF-MSCs.
Purpose: This study evaluated the chondrogenic potential of non-culture expanded synovium-mobilized MSCs and SF-microfragments obtained after enrichment using the STEM device and ascertained if device-mediated synovial membrane manipulation facilitated ongoing MSC release.
Study design: Controlled laboratory study.
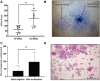
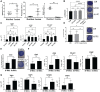
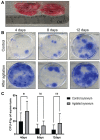
Methods: Two samples of aspiration fluid were collected intraoperatively before and after STEM device utilization from patients (n = 16) undergoing diagnostic or therapeutic knee arthroscopy. Human knee synovium (n = 5) was collected during total knee replacement, and a suspended culture was performed to assess the effect of the STEM device on ongoing MSC release. Colony forming unit-fibroblastic assays were used to determine the number of MSCs. Additionally, cytometric characterization of stromal and immune cells and chondrogenesis differentiation assay were performed without culture expansion. Filtered platelet concentrates were prepared using the HemaTrate system.
Results: After STEM device use, a significant increase was evident in SF-MSCs (P = .03) and synovial fluid-resident synovial tissue microfragments (P = .03). In vitro-suspended synovium released significantly more MSCs following STEM device use than nonstimulated synovium (P = .01). The STEM device-released total cellular fraction produced greater in vitro chondrogenesis with significantly more glycosaminoglycans (GAGs; P < .0001) when compared with non-STEM device synovial fluid material. Nonexpanded SF-MSCs and SF-microfragments combined with autologous filtered platelet concentrate produced significantly more GAGs than the complete chondrogenic media (P < .0001). The STEM device-mobilized cells contained more M2 macrophage cells and fewer M1 cells.
Conclusion: Non-culture expanded SF-MSCs and SF-microfragments had the potential to undergo chondrogenesis without culture expansion, which can be augmented using the STEM device with increased MSC release from manipulated synovium for several days. Although preliminary, these findings offer proof of concept toward manipulation of the knee joint environment to facilitate endogenous repair responses.
Clinical relevance: Although numbers were small, this study highlights 3 factors relevant to 1-stage joint repair using the STEM device: increased SF-MSCs and SF-microfragments and prolonged synovial release of MSCs. Joint repair strategies involving endogenous MSCs for cartilage repair without the need for culture expansion in a 1-stage procedure may be possible.
Keywords: nonexpanded cells; platelet products; synovial resident mesenchymal stem cells.
Conflict of interest statement
One or more of the authors has declared the following potential conflict of interest or source of funding: This article presents independent research funded/supported by the Leeds Biomedical Research Centre, National Institute for Health Research. H.P. is a senior investigator for the National Institute for Health Research. T.G.B., E.J., and D.M. hold a Patent US 2018 / 0161022 0161022 A1 for orthopaedic medical device. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Figures

Similar articles
-
A Novel Arthroscopic Technique for Intraoperative Mobilization of Synovial Mesenchymal Stem Cells.Am J Sports Med. 2018 Dec;46(14):3532-3540. doi: 10.1177/0363546518803757. Epub 2018 Nov 12. Am J Sports Med. 2018. PMID: 30419170 Free PMC article.
-
Platelet lysate enhances synovial fluid multipotential stromal cells functions: Implications for therapeutic use.Cytotherapy. 2018 Mar;20(3):375-384. doi: 10.1016/j.jcyt.2017.12.003. Epub 2018 Feb 3. Cytotherapy. 2018. PMID: 29398623
-
Human osteoarthritic synovium impacts chondrogenic differentiation of mesenchymal stem cells via macrophage polarisation state.Osteoarthritis Cartilage. 2014 Aug;22(8):1167-75. doi: 10.1016/j.joca.2014.05.021. Epub 2014 Jun 7. Osteoarthritis Cartilage. 2014. PMID: 24911520
-
Synovial membrane mesenchymal stem cells: past life, current situation, and application in bone and joint diseases.Stem Cell Res Ther. 2020 Sep 7;11(1):381. doi: 10.1186/s13287-020-01885-3. Stem Cell Res Ther. 2020. PMID: 32894205 Free PMC article. Review.
-
Synovial Fluid Mesenchymal Stem Cells for Knee Arthritis and Cartilage Defects: A Review of the Literature.J Knee Surg. 2021 Nov;34(13):1476-1485. doi: 10.1055/s-0040-1710366. Epub 2020 May 13. J Knee Surg. 2021. PMID: 32403148 Review.
Cited by
-
Body Fluid-Derived Stem Cells: Powering Innovative, Less-Invasive Cell Therapies.Int J Mol Sci. 2025 May 5;26(9):4382. doi: 10.3390/ijms26094382. Int J Mol Sci. 2025. PMID: 40362618 Free PMC article. Review.
References
-
- Akgun I, Unlu MC, Erdal OA, et al.. Matrix-induced autologous mesenchymal stem cell implantation versus matrix-induced autologous chondrocyte implantation in the treatment of chondral defects of the knee: a 2-year randomized study. Arch Orthop Trauma Surg. 2015;135(2):251-263. - PubMed
-
- Altaie A, Baboolal TG, Wall O, Jones E, McGonagle D. Platelet lysate enhances synovial fluid multipotential stromal cells functions: implications for therapeutic use. Cytotherapy. 2018;20(3):375-384. - PubMed
-
- Anitua E, Sánchez M, Nurden AT, et al.. Platelet-released growth factors enhance the secretion of hyaluronic acid and induce hepatocyte growth factor production by synovial fibroblasts from arthritic patients. Rheumatology (Oxford). 2007;46(12):1769-1772. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

