Identification and Mapping Real-World Data Sources for Heart Failure, Acute Coronary Syndrome, and Atrial Fibrillation
- PMID: 34781301
- PMCID: PMC8985014
- DOI: 10.1159/000520674
Identification and Mapping Real-World Data Sources for Heart Failure, Acute Coronary Syndrome, and Atrial Fibrillation
Abstract
Background: Transparent and robust real-world evidence sources are increasingly important for global health, including cardiovascular (CV) diseases. We aimed to identify global real-world data (RWD) sources for heart failure (HF), acute coronary syndrome (ACS), and atrial fibrillation (AF).
Methods: We conducted a systematic review of publications with RWD pertaining to HF, ACS, and AF (2010-2018), generating a list of unique data sources. Metadata were extracted based on the source type (e.g., electronic health records, genomics, and clinical data), study design, population size, clinical characteristics, follow-up duration, outcomes, and assessment of data availability for future studies and linkage.
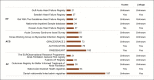
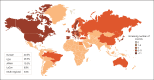
Results: Overall, 11,889 publications were retrieved for HF, 10,729 for ACS, and 6,262 for AF. From these, 322 (HF), 287 (ACS), and 220 (AF) data sources were selected for detailed review. The majority of data sources had near complete data on demographic variables (HF: 94%, ACS: 99%, and AF: 100%) and considerable data on comorbidities (HF: 77%, ACS: 93%, and AF: 97%). The least reported data categories were drug codes (HF, ACS, and AF: 10%) and caregiver involvement (HF: 6%, ACS: 1%, and AF: 1%). Only a minority of data sources provided information on access to data for other researchers (11%) or whether data could be linked to other data sources to maximize clinical impact (20%). The list and metadata for the RWD sources are publicly available at www.escardio.org/bigdata.
Conclusions: This review has created a comprehensive resource of CV data sources, providing new avenues to improve future real-world research and to achieve better patient outcomes.
Keywords: Cardiovascular; Data sources; Real-world data; Real-world evidence.
© 2021 The Author(s). Published by S. Karger AG, Basel.
Conflict of interest statement
Prof. Kotecha reports, outside of this study, grants from the National Institute for Health Research (NIHR CDF-2015-08-074 RATE-AF; NIHR HTA-130280 DaRe2THINK; NIHR EME-132974 D2T-NV), the British Heart Foundation (PG/17/55/33087 and AA/18/2/34218), EU/EFPIA IMI (BigData@Heart 116074), the European Society of Cardiology supported by educational grants from Boehringer Ingelheim/BMS-Pfizer Alliance/Bayer/Daiichi Sankyo/Boston Scientific, the NIHR/the University of Oxford Biomedical Research Centre, and British Heart Foundation/the University of Birmingham Accelerator Award (STEEER-AF NCT04396418), Amomed Pharma, and IRCCS San Raffaele/Menarini (beta-blockers in Heart Failure Collaborative Group NCT0083244), in addition to personal fees from Bayer (Advisory Board), AtriCure (Speaker fees), Protherics Medicines Development (Advisory Board), and Myokardia (Advisory Board). Prof. Kotecha is one of the associate editors to Cardiology. Dr. Sartini, Dr. Agrawal, Dr. Natani, Prof. Denaxas, Prof. Asselbergs, and Prof. Dobson have nothing to disclose. Dr. Gill reports funding through the BigData@Heart IMI, grant No. 116074. Dr. Suzart-Woischnik reports personal fees from Bayer AG, during the conduct of the study, and other from Bayer AG, outside the submitted work. Dr. Wirta reports other from Novartis, during the conduct of the study. Dr. Studer reports personal fees from Novartis Pharmaceuticals, during the conduct of the study, and other from Novartis Pharmaceuticals, outside the submitted work.
Figures



References
-
- World Health Organization Cardiovascular diseases (CVDs) 2020. Available from: http://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-...
-
- Wang H, Naghavi M, Allen C, Barber RM, Bhutta ZA, Carter A, et al. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: a systematic analysis for the global burden of disease study 2015. The Lancet. 2016;388((10053)):1459–544. - PMC - PubMed
-
- World Health Organisation . The future of CVD. WHO; 2020. Available from: https://www.who.int/cardiovascular_diseases/en/cvd_atlas_25_future.pdf?ua=1.
-
- World Health Organisation Global status report on noncommunicable diseases 2010: description of the global burden of NCDs, their risk factors and determinants. 2020. Available from: https://www.who.int/nmh/publications/ncd_report2010/en/
-
- U.S. Food & Drug Administration Real world evidence. 2020. Available from: https://www.fda.gov/science-research/science-and-research-special-topics....
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

