Cost-effectiveness of eltrombopag vs intravenous immunoglobulin for the perioperative management of immune thrombocytopenia
- PMID: 34781363
- PMCID: PMC8945289
- DOI: 10.1182/bloodadvances.2021005627
Cost-effectiveness of eltrombopag vs intravenous immunoglobulin for the perioperative management of immune thrombocytopenia
Abstract
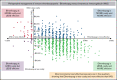
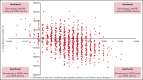
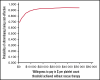
Eltrombopag has been shown to be noninferior to intravenous immunoglobulin (IVIG) for improving perioperative platelet counts in patients with immune thrombocytopenia (ITP) in a randomized trial; thus, cost is an important factor for treatment and policy decisions. We used patient-level data from the trial to conduct a cost-effectiveness analysis comparing perioperative eltrombopag 50 mg daily starting dose, with IVIG 1 or 2 g/kg (according to local practice) from a Canadian public health care payer's perspective over the observation period, from preoperative day 21 to postoperative day 28. Resource utilization data were obtained from the trial data (eltrombopag, n = 38; IVIG, n = 36), and unit costs were collected from the Ontario Schedule of Benefits, Ontario Drug Formulary, and secondary sources. All costs were adjusted to 2020 Canadian dollars. We calculated the incremental cost per patient for all patients randomized. Uncertainty was addressed using nonparametric bootstrapping. The use of perioperative eltrombopag for patients with ITP resulted in a cost-saving of $413 Canadian per patient. Compared with IVIG, the probability of eltrombopag being cost effective was 70% even with no willingness to pay. In a sensitivity analysis based on IVIG dose, we found that with the higher dose of IVIG (2 g/kg), eltrombopag saved $2,714 per patient, whereas with the lower dose of IVIG (1 g/kg), eltrombopag had a higher mean cost of $562 per patient. In summary, based on data from the randomized trial that demonstrated noninferiority, the use of eltrombopag for the management of ITP in the perioperative setting was less costly than IVIG.
© 2022 by The American Society of Hematology. Licensed under Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0), permitting only noncommercial, nonderivative use with attribution. All other rights reserved.
Figures

References
-
- Provan D, Stasi R, Newland AC, et al. . International consensus report on the investigation and management of primary immune thrombocytopenia. Blood. 2010;115(2):168-186. - PubMed
-
- Cines DB, Blanchette VS. Immune thrombocytopenic purpura. N Engl J Med. 2002;346(13):995-1008. - PubMed
-
- Glance LG, Dick AW, Mukamel DB, et al. . Association between intraoperative blood transfusion and mortality and morbidity in patients undergoing noncardiac surgery. Anesthesiology. 2011;114(2):283-292. - PubMed

