A large panel of chicken cells are invaded in vivo by Salmonella Typhimurium even when depleted of all known invasion factors
- PMID: 34784793
- PMCID: PMC8596019
- DOI: 10.1098/rsob.210117
A large panel of chicken cells are invaded in vivo by Salmonella Typhimurium even when depleted of all known invasion factors
Abstract
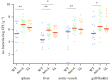
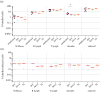
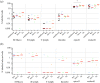
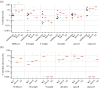
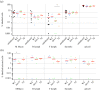
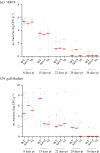
Poultry are the main source of human infection by Salmonella. As infected poultry are asymptomatic, identifying infected poultry farms is difficult, thus controlling animal infections is of primary importance. As cell tropism is known to govern disease, our aim was therefore to identify infected host-cell types in the organs of chicks known to be involved in Salmonella infection and investigate the role of the three known invasion factors in this process (T3SS-1, Rck and PagN). Chicks were inoculated with wild-type or isogenic fluorescent Salmonella Typhimurium mutants via the intracoelomic route. Our results show that liver, spleen, gall bladder and aortic vessels could be foci of infection, and that phagocytic and non-phagocytic cells, including immune, epithelial and endothelial cells, are invaded in vivo in each organ. Moreover, a mutant defective for the T3SS-1, Rck and PagN remained able to colonize organs like the wild-type strain and invaded non-phagocytic cells in each organ studied. As the infection of the gall bladder had not previously been described in chicks, invasion of gall bladder cells was confirmed by immunohistochemistry and infection was shown to last several weeks after inoculation. Altogether, for the first time these findings provide insights into cell tropism of Salmonella in relevant organs involved in Salmonella infection in chicks and also demonstrate that the known invasion factors are not required for entry into these cell types.
Keywords: Salmonella; gall bladder; host–pathogen interaction; invasion; poultry.
Conflict of interest statement
We declare we have no competing interests.
Figures


Similar articles
-
Salmonella Typhimurium Invalidated for the Three Currently Known Invasion Factors Keeps Its Ability to Invade Several Cell Models.Front Cell Infect Microbiol. 2018 Aug 10;8:273. doi: 10.3389/fcimb.2018.00273. eCollection 2018. Front Cell Infect Microbiol. 2018. PMID: 30148118 Free PMC article.
-
TolC but not AcrB is essential for multidrug-resistant Salmonella enterica serotype Typhimurium colonization of chicks.J Antimicrob Chemother. 2005 May;55(5):707-12. doi: 10.1093/jac/dki091. Epub 2005 Apr 6. J Antimicrob Chemother. 2005. PMID: 15814604
-
Short-term feeding of probiotics and synbiotics modulates caecal microbiota during Salmonella Typhimurium infection but does not reduce shedding and invasion in chickens.Appl Microbiol Biotechnol. 2020 Jan;104(1):319-334. doi: 10.1007/s00253-019-10220-7. Epub 2019 Nov 22. Appl Microbiol Biotechnol. 2020. PMID: 31758235
-
The Salmonella pathogenicity island 1 and Salmonella pathogenicity island 2 type III secretion systems play a major role in pathogenesis of systemic disease and gastrointestinal tract colonization of Salmonella enterica serovar Typhimurium in the chicken.Avian Pathol. 2007 Jun;36(3):199-203. doi: 10.1080/03079450701264118. Avian Pathol. 2007. PMID: 17497331
-
Evaluation of respiratory route as a viable portal of entry for Salmonella in poultry.Vet Med (Auckl). 2014 Aug 14;5:59-73. doi: 10.2147/VMRR.S62775. eCollection 2014. Vet Med (Auckl). 2014. PMID: 32670847 Free PMC article. Review.
Cited by
-
High Affinity Iron Acquisition Systems Facilitate but Are Not Essential for Colonization of Chickens by Salmonella Enteritidis.Front Microbiol. 2022 Mar 3;13:824052. doi: 10.3389/fmicb.2022.824052. eCollection 2022. Front Microbiol. 2022. PMID: 35308377 Free PMC article.
-
Synergistic influence of probiotic and florfenicol on embryonic viability, performance, and multidrug-resistant Salmonella Enteritidis in broiler chickens.Sci Rep. 2023 Jun 14;13(1):9644. doi: 10.1038/s41598-023-36238-6. Sci Rep. 2023. PMID: 37316527 Free PMC article.
-
The Salmonella virulence protein PagN contributes to the advent of a hyper-replicating cytosolic bacterial population.Virulence. 2024 Dec;15(1):2357670. doi: 10.1080/21505594.2024.2357670. Epub 2024 May 28. Virulence. 2024. PMID: 38804638 Free PMC article.
References
-
- WHO. 2017. WHO estimates of the global burden of foodborne diseases. See https://apps.who.int/iris/handle/10665/199350.
-
- Rabinowitz PM, Conti LA. 2009. Human-clinical-medicine: clinical approaches to zoonoses, toxicants and other shared health risks. 1st edn. Maryland Heights, MD: Saunders.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

