Optimal fludarabine lymphodepletion is associated with improved outcomes after CAR T-cell therapy
- PMID: 34788386
- PMCID: PMC9006295
- DOI: 10.1182/bloodadvances.2021006418
Optimal fludarabine lymphodepletion is associated with improved outcomes after CAR T-cell therapy
Erratum in
-
Fabrizio VA, Boelens JJ, Mauguen A, et al. Optimal fludarabine lymphodepletion is associated with improved outcomes after CAR T-cell therapy. Blood Adv. 2022;6(7):1961-1968.Blood Adv. 2023 Jun 27;7(12):2924. doi: 10.1182/bloodadvances.2023010269. Blood Adv. 2023. PMID: 37368453 Free PMC article. No abstract available.
Abstract
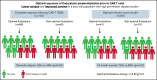
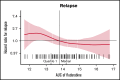
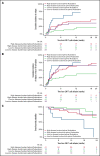
Chimeric antigen receptor (CAR) T cells provide a therapeutic option in hematologic malignancies. However, treatment failure after initial response approaches 50%. In allogeneic hematopoietic cell transplantation, optimal fludarabine exposure improves immune reconstitution, resulting in lower nonrelapse mortality and increased survival. We hypothesized that optimal fludarabine exposure in lymphodepleting chemotherapy before CAR T-cell therapy would improve outcomes. In a retrospective analysis of patients with relapsed/refractory B-cell acute lymphoblastic leukemia undergoing CAR T-cell (tisagenlecleucel) infusion after cyclophosphamide/fludarabine lymphodepleting chemotherapy, we estimated fludarabine exposure as area under the curve (AUC; mg × h/L) using a validated population pharmacokinetic (PK) model. Fludarabine exposure was related to overall survival (OS), cumulative incidence of relapse (CIR), and a composite end point (loss of B-cell aplasia [BCA] or relapse). Eligible patients (n = 152) had a median age of 12.5 years (range, <1 to 26), response rate of 86% (n = 131 of 152), 12-month OS of 75.1% (95% confidence interval [CI], 67.6% to 82.6%), and 12-month CIR of 36.4% (95% CI, 27.5% to 45.2%). Optimal fludarabine exposure was determined as AUC ≥13.8 mg × h/L. In multivariable analyses, patients with AUC <13.8 mg × h/L had a 2.5-fold higher CIR (hazard ratio [HR], 2.45; 95% CI, 1.34-4.48; P = .005) and twofold higher risk of relapse or loss of BCA (HR, 1.96; 95% CI, 1.19-3.23; P = .01) compared with those with optimal fludarabine exposure. High preinfusion disease burden was also associated with increased risk of relapse (HR, 2.66; 95% CI, 1.45-4.87; P = .001) and death (HR, 4.77; 95% CI, 2.10-10.9; P < .001). Personalized PK-directed dosing to achieve optimal fludarabine exposure should be tested in prospective trials and, based on this analysis, may reduce disease relapse after CAR T-cell therapy.
© 2022 by The American Society of Hematology. Licensed under Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0), permitting only noncommercial, nonderivative use with attribution. All other rights reserved.
Figures
References
-
- Gaynon PS. Childhood acute lymphoblastic leukaemia and relapse. Br J Haematol. 2005;131(5):579-587. - PubMed
-
- von Stackelberg A, Völzke E, Kühl JS, et al. ; ALL-REZ BFM Study Group . Outcome of children and adolescents with relapsed acute lymphoblastic leukaemia and non-response to salvage protocol therapy: a retrospective analysis of the ALL-REZ BFM Study Group. Eur J Cancer. 2011;47(1):90-97. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

