Deletion of p38γ attenuates ethanol consumption- and acetaminophen-induced liver injury in mice through promoting Dlg1
- PMID: 34789918
- PMCID: PMC9253030
- DOI: 10.1038/s41401-021-00795-1
Deletion of p38γ attenuates ethanol consumption- and acetaminophen-induced liver injury in mice through promoting Dlg1
Abstract
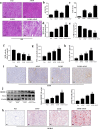
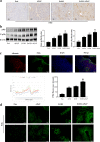
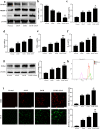
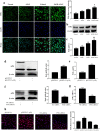
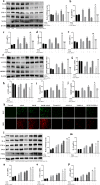
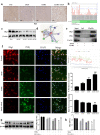
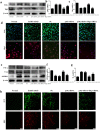
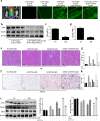
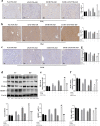
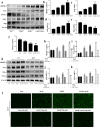
Acetaminophen (APAP) is one of the major causes of drug-induced acute liver injury, and ethanol may aggravate APAP-induced liver injury. The problem of ethanol- and APAP-induced liver injury becomes increasingly prominent, but the mechanism of ethanol- and APAP-induced liver injury remains ambiguous. p38γ is one of the four isoforms of P38 mitogen activated protein kinases, that contributes to inflammation in different diseases. In this study we investigated the role of p38γ in ethanol- and APAP-induced liver injury. Liver injury was induced in male C57BL/6 J mice by giving liquid diet containing 5% ethanol (v/v) for 10 days, followed by gavage of ethanol (25% (v/v), 6 g/kg) once or injecting APAP (200 mg/kg, ip), or combined the both treatments. We showed that ethanol significantly aggravated APAP-induced liver injury in C57BL/6 J mice. Moreover, the expression level of p38γ was up-regulated in the liver of ethanol-, APAP- and ethanol+APAP-treated mice. Knockdown of p38γ markedly attenuated liver injury, inflammation, and steatosis in ethanol+APAP-treated mice. Liver sections of p38γ-knockdown mice displayed lower levels of Oil Red O stained dots and small leaky shapes. AML-12 cells were exposed to APAP (5 mM), ethanol (100 mM) or combined treatments. We showed that P38γ was markedly increased in ethanol+APAP-treated AML-12 cells, whereas knockdown of p38γ significantly inhibited inflammation, lipid accumulation and oxidative stress in ethanol+APAP-treated AML-12 cells. Furthermore, we revealed that p38γ could combine with Dlg1, a member of membrane-associated guanylate kinase family. Deletion of p38γ up-regulated the expression level of Dlg1 in ethanol+APAP-treated AML-12 cells. In summary, our results suggest that p38γ functions as an important regulator in ethanol- and APAP-induced liver injury through modulation of Dlg1.
Keywords: Dlg1; P38γ; acetaminophen; ethanol; fatty liver; steatohepatitis.
© 2021. The Author(s), under exclusive licence to CPS and SIMM.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
P38γ modulates the lipid metabolism in non-alcoholic fatty liver disease by regulating the JAK-STAT signaling pathway.FASEB J. 2023 Jan;37(1):e22716. doi: 10.1096/fj.202200939RR. FASEB J. 2023. PMID: 36527390
-
Ethanol Extract of Rosa rugosa Ameliorates Acetaminophen-Induced Liver Injury via Upregulating Sirt1 and Subsequent Potentiation of LKB1/AMPK/Nrf2 Cascade in Hepatocytes.Molecules. 2023 Oct 28;28(21):7307. doi: 10.3390/molecules28217307. Molecules. 2023. PMID: 37959727 Free PMC article.
-
Caveolin-1 alleviates acetaminophen-induced vascular oxidative stress and inflammation in non-alcoholic fatty liver disease.Free Radic Biol Med. 2023 Feb 1;195:245-257. doi: 10.1016/j.freeradbiomed.2022.12.095. Epub 2022 Dec 31. Free Radic Biol Med. 2023. PMID: 36596386
-
Chitinase 3-like-1 contributes to acetaminophen-induced liver injury by promoting hepatic platelet recruitment.Elife. 2021 Jun 10;10:e68571. doi: 10.7554/eLife.68571. Elife. 2021. PMID: 34110284 Free PMC article.
-
Mitoquinone protects against acetaminophen-induced liver injury in an FSP1-dependent and GPX4-independent manner.Toxicol Appl Pharmacol. 2023 Apr 15;465:116452. doi: 10.1016/j.taap.2023.116452. Epub 2023 Mar 7. Toxicol Appl Pharmacol. 2023. PMID: 36894071
Cited by
-
Advancements in the Alcohol-Associated Liver Disease Model.Biomolecules. 2022 Jul 27;12(8):1035. doi: 10.3390/biom12081035. Biomolecules. 2022. PMID: 36008929 Free PMC article. Review.
-
Modulation of Paracetamol-Induced Hepatotoxicity by Acute and Chronic Ethanol Consumption in Mice: A Study Pilot.Toxics. 2024 Nov 27;12(12):857. doi: 10.3390/toxics12120857. Toxics. 2024. PMID: 39771072 Free PMC article.
-
Salvianolic Acid A Protects against Acetaminophen-Induced Hepatotoxicity via Regulation of the miR-485-3p/SIRT1 Pathway.Antioxidants (Basel). 2023 Apr 3;12(4):870. doi: 10.3390/antiox12040870. Antioxidants (Basel). 2023. PMID: 37107244 Free PMC article.
-
Studies on the Changes of Fermentation Metabolites and the Protective Effect of Fermented Edible Grass on Stress Injury Induced by Acetaminophen in HepG2 Cells.Foods. 2024 Feb 2;13(3):470. doi: 10.3390/foods13030470. Foods. 2024. PMID: 38338605 Free PMC article.
-
Label-Free Imaging Techniques to Evaluate Metabolic Changes Caused by Toxic Liver Injury in PCLS.Int J Mol Sci. 2023 May 24;24(11):9195. doi: 10.3390/ijms24119195. Int J Mol Sci. 2023. PMID: 37298155 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

