Emerging agents that target signaling pathways to eradicate colorectal cancer stem cells
- PMID: 34791817
- PMCID: PMC8696218
- DOI: 10.1002/cac2.12235
Emerging agents that target signaling pathways to eradicate colorectal cancer stem cells
Abstract
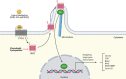
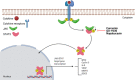
Colorectal cancer (CRC) represents the third most commonly diagnosed cancer and the second leading cause of cancer death worldwide. The modern concept of cancer biology indicates that cancer is formed of a small population of cells called cancer stem cells (CSCs), which present both pluripotency and self-renewal properties. These cells are considered responsible for the progression of the disease, recurrence and tumor resistance. Interestingly, some cell signaling pathways participate in CRC survival, proliferation, and self-renewal properties, and most of them are dysregulated in CSCs, including the Wingless (Wnt)/β-catenin, Notch, Hedgehog, nuclear factor kappa B (NF-κB), Janus kinase/signal transducer and activator of transcription (JAK/STAT), peroxisome proliferator-activated receptor (PPAR), phosphatidyl-inositol-3-kinase/Akt/mechanistic target of rapamycin (PI3K/Akt/mTOR), and transforming growth factor-β (TGF-β)/Smad pathways. In this review, we summarize the strategies for eradicating CRC stem cells by modulating these dysregulated pathways, which will contribute to the study of potential therapeutic schemes, combining conventional drugs with CSC-targeting drugs, and allowing better cure rates in anti-CRC therapy.
Keywords: Hedgehog; JAK/STAT signaling; NF-κB; Notch; PI3K/Akt/mTOR signaling; Wnt/β-catenin pathway; cancer stem cells; cell signaling; colorectal; targeted therapy.
© 2021 The Authors. Cancer Communications published by John Wiley & Sons Australia, Ltd. on behalf of Sun Yat-sen University Cancer Center.
Conflict of interest statement
The authors have declared that there are no conflicts of interest.
Figures






References
-
- Giacchetti S, Perpoint B, Zidani R, Le Bail N, Faggiuolo R, Focan C, et al. Phase III multicenter randomized trial of oxaliplatin added to chronomodulated fluorouracil‐leucovorin as first‐line treatment of metastatic colorectal cancer. J Clin Oncol. 2000;18:136–47. 10.1200/JCO.2000.18.1.136. - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

