Peripherally-driven myeloid NFkB and IFN/ISG responses predict malignancy risk, survival, and immunotherapy regime in ovarian cancer
- PMID: 34795003
- PMCID: PMC8603275
- DOI: 10.1136/jitc-2021-003609
Peripherally-driven myeloid NFkB and IFN/ISG responses predict malignancy risk, survival, and immunotherapy regime in ovarian cancer
Abstract
Background: Tumors can influence peripheral immune macroenvironment, thereby creating opportunities for non-invasive serum/plasma immunobiomarkers for immunostratification and immunotherapy designing. However, current approaches for immunobiomarkers' detection are largely quantitative, which is unreliable for assessing functional peripheral immunodynamics of patients with cancer. Hence, we aimed to design a functional biomarker modality for capturing peripheral immune signaling in patients with cancer for reliable immunostratification.
Methods: We used a data-driven in silico framework, integrating existing tumor/blood bulk-RNAseq or single-cell (sc)RNAseq datasets of patients with cancer, to inform the design of an innovative serum-screening modality, that is, serum-functional immunodynamic status (sFIS) assay. Next, we pursued proof-of-concept analyses via multiparametric serum profiling of patients with ovarian cancer (OV) with sFIS assay combined with Luminex (cytokines/soluble immune checkpoints), CA125-antigen detection, and whole-blood immune cell counts. Here, sFIS assay's ability to determine survival benefit or malignancy risk was validated in a discovery (n=32) and/or validation (n=699) patient cohorts. Lastly, we used an orthotopic murine metastatic OV model, with anti-OV therapy selection via in silico drug-target screening and murine serum screening via sFIS assay, to assess suitable in vivo immunotherapy options.
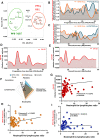
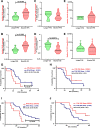
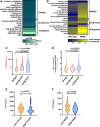
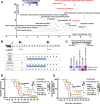
Results: In silico data-driven framework predicted that peripheral immunodynamics of patients with cancer might be best captured via analyzing myeloid nuclear factor kappa-light-chain enhancer of activated B cells (NFκB) signaling and interferon-stimulated genes' (ISG) responses. This helped in conceptualization of an 'in sitro' (in vitro+in situ) sFIS assay, where human myeloid cells were exposed to patients' serum in vitro, to assess serum-induced (si)-NFκB or interferon (IFN)/ISG responses (as active signaling reporter activity) within them, thereby 'mimicking' patients' in situ immunodynamic status. Multiparametric serum profiling of patients with OV established that sFIS assay can: decode peripheral immunology (by indicating higher enrichment of si-NFκB over si-IFN/ISG responses), estimate survival trends (si-NFκB or si-IFN/ISG responses associating with negative or positive prognosis, respectively), and coestimate malignancy risk (relative to benign/borderline ovarian lesions). Biologically, we documented dominance of pro-tumorigenic, myeloid si-NFκB responseHIGHsi-IFN/ISG responseLOW inflammation in periphery of patients with OV. Finally, in an orthotopic murine metastatic OV model, sFIS assay predicted the higher capacity of chemo-immunotherapy (paclitaxel-carboplatin plus anti-TNF antibody combination) in achieving a pro-immunogenic peripheral milieu (si-IFN/ISG responseHIGHsi-NFκB responseLOW), which aligned with high antitumor efficacy.
Conclusions: We established sFIS assay as a novel biomarker resource for serum screening in patients with OV to evaluate peripheral immunodynamics, patient survival trends and malignancy risk, and to design preclinical chemo-immunotherapy strategies.
Keywords: computational biology; immunological techniques; immunotherapy; tumor biomarkers; tumor microenvironment.
© Author(s) (or their employer(s)) 2021. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: The sFIS assay is currently the subject of an ongoing PCT patent application. ADG has received consulting/advisory/lecture honoraria from Boehringer Ingelheim and Miltenyi Biotec.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
