Ugi Reaction Synthesis of Oxindole-Lactam Hybrids as Selective Butyrylcholinesterase Inhibitors
- PMID: 34795859
- PMCID: PMC8591717
- DOI: 10.1021/acsmedchemlett.1c00344
Ugi Reaction Synthesis of Oxindole-Lactam Hybrids as Selective Butyrylcholinesterase Inhibitors
Abstract
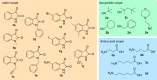
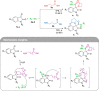
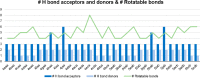
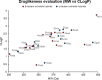
Molecular hybridization is a valuable approach in drug discovery. Combining it with multicomponent reactions is highly desirable, since structurally diverse libraries can be attained efficiently in an eco-friendly manner. In this work, isatin is used as the key building block for the Ugi 4-center 3-component reaction synthesis of oxindole-lactam hybrids, under catalyst-free conditions. The resulting oxindole-β-lactam and oxindole-γ-lactam hybrids were evaluated for their potential to inhibit relevant central nervous system targets, namely cholinesterases and monoamine oxidases. Druglikeness evaluation was also performed, and compounds 4eca and 5dab exhibited great potential as selective butyrylcholinesterase inhibitors, at the low micromolar range, with an interesting predictive pharmacokinetic profile. Our findings herein reported suggest oxindole-lactam hybrids as new potential agents for the treatment of Alzheimer's disease.
© 2021 American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



Similar articles
-
The Ugi4CR as effective tool to access promising anticancer isatin-based α-acetamide carboxamide oxindole hybrids.Beilstein J Org Chem. 2024 May 27;20:1213-1220. doi: 10.3762/bjoc.20.104. eCollection 2024. Beilstein J Org Chem. 2024. PMID: 38887573 Free PMC article.
-
One step access to oxindole-based β-lactams through Ugi four-center three-component reaction.RSC Adv. 2018 Oct 11;8(61):34903-34910. doi: 10.1039/c8ra08165d. eCollection 2018 Oct 10. RSC Adv. 2018. PMID: 35547060 Free PMC article.
-
The application of isatin-based multicomponent-reactions in the quest for new bioactive and druglike molecules.Eur J Med Chem. 2021 Feb 5;211:113102. doi: 10.1016/j.ejmech.2020.113102. Epub 2020 Dec 18. Eur J Med Chem. 2021. PMID: 33421712 Review.
-
Novel tacrine-grafted Ugi adducts as multipotent anti-Alzheimer drugs: a synthetic renewal in tacrine-ferulic acid hybrids.ChemMedChem. 2015 Mar;10(3):523-39. doi: 10.1002/cmdc.201402409. Epub 2014 Dec 23. ChemMedChem. 2015. PMID: 25537267
-
β-Lactams as promising anticancer agents: Molecular hybrids, structure activity relationships and potential targets.Eur J Med Chem. 2020 Sep 1;201:112510. doi: 10.1016/j.ejmech.2020.112510. Epub 2020 Jun 23. Eur J Med Chem. 2020. PMID: 32592915 Review.
Cited by
-
Multicomponent reactions driving the discovery and optimization of agents targeting central nervous system pathologies.Beilstein J Org Chem. 2024 Dec 3;20:3151-3173. doi: 10.3762/bjoc.20.261. eCollection 2024. Beilstein J Org Chem. 2024. PMID: 39669443 Free PMC article. Review.
-
Recent developments on microwave-assisted organic synthesis of nitrogen- and oxygen-containing preferred heterocyclic scaffolds.RSC Adv. 2023 Nov 7;13(47):32858-32892. doi: 10.1039/d3ra05986c. eCollection 2023 Nov 7. RSC Adv. 2023. PMID: 37942237 Free PMC article. Review.
-
Medicinal Chemistry in Portugal and Spain: A Strong Iberian Alliance.ACS Med Chem Lett. 2022 Jun 9;13(6):871-872. doi: 10.1021/acsmedchemlett.2c00194. eCollection 2022 Jun 9. ACS Med Chem Lett. 2022. PMID: 35707154 Free PMC article. No abstract available.
-
Ugi Four-Component Reactions Using Alternative Reactants.Molecules. 2023 Feb 8;28(4):1642. doi: 10.3390/molecules28041642. Molecules. 2023. PMID: 36838630 Free PMC article. Review.
-
The Ugi4CR as effective tool to access promising anticancer isatin-based α-acetamide carboxamide oxindole hybrids.Beilstein J Org Chem. 2024 May 27;20:1213-1220. doi: 10.3762/bjoc.20.104. eCollection 2024. Beilstein J Org Chem. 2024. PMID: 38887573 Free PMC article.
References
-
- Cioc R. C.; Ruijter E.; Orru R. V. A. Multicomponent reactions: advanced tools for sustainable organic synthesis. Green Chem. 2014, 16 (6), 2958–2975. 10.1039/C4GC00013G. - DOI
LinkOut - more resources
Full Text Sources
Chemical Information

