Implementing Digital-Waveform Technology for Extended m/ z Range Operation on a Native Dual-Quadrupole FT-IM-Orbitrap Mass Spectrometer
- PMID: 34797072
- PMCID: PMC9026758
- DOI: 10.1021/jasms.1c00245
Implementing Digital-Waveform Technology for Extended m/ z Range Operation on a Native Dual-Quadrupole FT-IM-Orbitrap Mass Spectrometer
Abstract
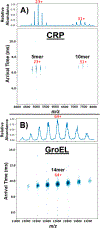
Here, we describe a digital-waveform dual-quadrupole mass spectrometer that enhances the performance of our drift tube FT-IMS high-resolution Orbitrap mass spectrometer (MS). The dual-quadrupole analyzer enhances the instrument capabilities for studies of large protein and protein complexes. The first quadrupole (q) provides a means for performing low-energy collisional activation of ions to reduce or eliminate noncovalent adducts, viz., salts, buffers, detergents, and/or endogenous ligands. The second quadrupole (Q) is used to mass-select ions of interest for further interrogation by ion mobility spectrometry and/or collision-induced dissociation (CID). Q is operated using digital-waveform technology (DWT) to improve the mass selection compared to that achieved using traditional sinusoidal waveforms at floated DC potentials (>500 V DC). DWT allows for increased precision of the waveform for a fraction of the cost of conventional RF drivers and with readily programmable operation and precision (Hoffman, N. M. . A comparison-based digital-waveform generator for high-resolution duty cycle. Review of Scientific Instruments 2018, 89, 084101).
Keywords: Fourier-transform ion mobility-Orbitrap mass spectrometry; digital-waveform technology; native mass spectrometry; quadrupole mass spectrometry.
Conflict of interest statement
The authors declare the following competing financial interest(s): The power supplies used for this instrument were purchased from G.A.A. Custom Electronics, owned by a co-author (G.A.A.). Other authors declare no competing financial interests.
Figures




References
-
- Clemmer DE; Russell DH; Williams ER Characterizing the Conformationome: Toward a Structural Understanding of the Proteome. Acc. Chem. Res 2017, 50 (3), 556–560. - PubMed
-
- Bai X-C; McMullan G; Scheres SHW How cryo-EM is revolutionizing structural biology. Trends Biochem. Sci 2015, 40 (1), 49–57. - PubMed
-
- Murata K; Wolf M Cryo-electron microscopy for structural analysis of dynamic biological macromolecules. Biochim. Biophys. Acta, Gen. Subj 2018, 1862 (2), 324–334. - PubMed
-
- Shi Y A Glimpse of Structural Biology through X-Ray Crystallography. Cell 2014, 159 (5), 995–1014. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

