Safety and efficacy of intermittent presumptive treatment with sulfadoxine-pyrimethamine using rapid diagnostic test screening and treatment with dihydroartemisinin-piperaquine at the first antenatal care visit (IPTp-SP+): study protocol for a randomized controlled trial
- PMID: 34801059
- PMCID: PMC8605457
- DOI: 10.1186/s13063-021-05745-0
Safety and efficacy of intermittent presumptive treatment with sulfadoxine-pyrimethamine using rapid diagnostic test screening and treatment with dihydroartemisinin-piperaquine at the first antenatal care visit (IPTp-SP+): study protocol for a randomized controlled trial
Abstract
Background: Intermittent preventive treatment in pregnancy (IPTp) with sulfadoxine-pyrimethamine (SP) is recommended by the World Health Organization for the prevention of malaria in pregnancy (MIP)-associated adverse outcomes in high burden areas. However, the efficacy of IPTp-SP has decreased in step with increasing parasite drug resistance. Suitable alternative strategies are needed.
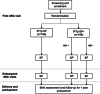
Methods: This is a protocol for a phase IIIb open-label, two-armed randomized controlled superiority trial to assess the safety and efficacy of a hybrid approach to IPTp combining screening and treatment with dihydroartemisinin-piperaquine (DP) to the current IPTp-SP regimen at the first antenatal care clinic visit. Pregnant women without HIV infection and without signs or symptoms of malaria will be randomized to either standard IPTp-SP or hybrid IPTp-SP plus screening and treatment (IPTp-SP+). In the IPTp-SP+ arm, participants who screen positive by rapid diagnostic test for P. falciparum will be treated with DP at the first antenatal visit while those who screen negative will receive SP per current guidelines. All participants will be administered SP on days 35 and 63 and will be actively followed biweekly up to day 63 and then monthly until delivery. Infants will be followed until 1 year after delivery. The primary endpoint is incident PCR-confirmed MIP at day 42. Secondary endpoints include incident MIP at other time points, placental malaria, congenital malaria, hemoglobin trends, birth outcomes, and incidence of adverse events in infants up to the first birthday.
Discussion: A hybrid approach to IPTp that combines screening and treatment with an artemisinin-based combination therapy at the first visit with standard IPTp-SP is hypothesized to confer added benefit over IPTp-SP alone in a high malaria transmission area with prevalent SP resistant parasites.
Trial registration: Pan African Clinical Trials Registry 201905721140808 . Registered retrospectively on 11 May 2019.
Keywords: Dihydroartemisinin-piperaquine; Intermittent presumptive therapy; Malaria in pregnancy; Sulfadoxine-pyrimethamine; Zambia.
© 2021. The Author(s).
Conflict of interest statement
JBK is an EDCTP fellow. The other authors declare that they have no competing interests.
Figures
References
-
- World Health Organization . World Malaria Report. Geneva: WHO Global Malaria Programme; 2020.
-
- World Health Organization . A strategic framework for malaria prevention and control during pregnancy in the African region. Geneva: WHO; 2004.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical