Hippocampal Function Is Impaired by a Short-Term High-Fat Diet in Mice: Increased Blood-Brain Barrier Permeability and Neuroinflammation as Triggering Events
- PMID: 34803583
- PMCID: PMC8600238
- DOI: 10.3389/fnins.2021.734158
Hippocampal Function Is Impaired by a Short-Term High-Fat Diet in Mice: Increased Blood-Brain Barrier Permeability and Neuroinflammation as Triggering Events
Abstract
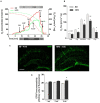
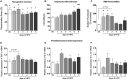
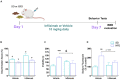
Worldwide, and especially in Western civilizations, most of the staple diets contain high amounts of fat and refined carbohydrates, leading to an increasing number of obese individuals. In addition to inducing metabolic disorders, energy dense food intake has been suggested to impair brain functions such as cognition and mood control. Here we demonstrate an impaired memory function already 3 days after the start of a high-fat diet (HFD) exposure, and depressive-like behavior, in the tail suspension test, after 5 days. These changes were followed by reduced synaptic density, changes in mitochondrial function and astrocyte activation in the hippocampus. Preceding or coinciding with the behavioral changes, we found an induction of the proinflammatory cytokines TNF-α and IL-6 and an increased permeability of the blood-brain barrier (BBB), in the hippocampus. Finally, in mice treated with a TNF-α inhibitor, the behavioral and BBB alterations caused by HFD-feeding were mitigated suggesting that inflammatory signaling was critical for the changes. In summary, our findings suggest that HFD rapidly triggers hippocampal dysfunction associated with BBB disruption and neuroinflammation, promoting a progressive breakdown of synaptic and metabolic function. In addition to elucidating the link between diet and cognitive function, our results might be relevant for the comprehension of the neurodegenerative process.
Keywords: bioenergetics; blood–brain barrier; cognition; depression; high fat diet; memory; mitochondria; neuroinflammation.
Copyright © 2021 de Paula, Brunetta, Engel, Gaspar, Velloso, Engblom, de Oliveira and de Bem.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures