Emerging Role of Non-Coding RNAs in Regulation of T-Lymphocyte Function
- PMID: 34804042
- PMCID: PMC8599985
- DOI: 10.3389/fimmu.2021.756042
Emerging Role of Non-Coding RNAs in Regulation of T-Lymphocyte Function
Abstract
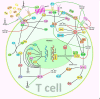
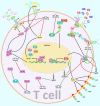
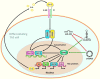
T-lymphocytes (T cells) play a major role in adaptive immunity and current immune checkpoint inhibitor-based cancer treatments. The regulation of their function is complex, and in addition to cytokines, receptors and transcription factors, several non-coding RNAs (ncRNAs) have been shown to affect differentiation and function of T cells. Among these non-coding RNAs, certain small microRNAs (miRNAs) including miR-15a/16-1, miR-125b-5p, miR-99a-5p, miR-128-3p, let-7 family, miR-210, miR-182-5p, miR-181, miR-155 and miR-10a have been well recognized. Meanwhile, IFNG-AS1, lnc-ITSN1-2, lncRNA-CD160, NEAT1, MEG3, GAS5, NKILA, lnc-EGFR and PVT1 are among long non-coding RNAs (lncRNAs) that efficiently influence the function of T cells. Recent studies have underscored the effects of a number of circular RNAs, namely circ_0001806, hsa_circ_0045272, hsa_circ_0012919, hsa_circ_0005519 and circHIPK3 in the modulation of T-cell apoptosis, differentiation and secretion of cytokines. This review summarizes the latest news and regulatory roles of these ncRNAs on the function of T cells, with widespread implications on the pathophysiology of autoimmune disorders and cancer.
Keywords: T cell; autoimmune; cancer; circRNA; lncRNA; miRNA.
Copyright © 2021 Taheri, Barth, Kargl, Rezaei, Ghafouri-Fard and Pichler.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Regulatory Role of Non-Coding RNAs on Immune Responses During Sepsis.Front Immunol. 2021 Dec 9;12:798713. doi: 10.3389/fimmu.2021.798713. eCollection 2021. Front Immunol. 2021. PMID: 34956235 Free PMC article. Review.
-
The key roles of non-coding RNAs in the pathophysiology of hypertension.Eur J Pharmacol. 2022 Sep 15;931:175220. doi: 10.1016/j.ejphar.2022.175220. Epub 2022 Aug 19. Eur J Pharmacol. 2022. PMID: 35995213 Review.
-
Non-Coding RNAs in the Etiology and Control of Major and Neglected Human Tropical Diseases.Front Immunol. 2021 Oct 19;12:703936. doi: 10.3389/fimmu.2021.703936. eCollection 2021. Front Immunol. 2021. PMID: 34737736 Free PMC article. Review.
-
The Emerging Role of Non-Coding RNAs in Pituitary Gland Tumors and Meningioma.Cancers (Basel). 2021 Nov 28;13(23):5987. doi: 10.3390/cancers13235987. Cancers (Basel). 2021. PMID: 34885097 Free PMC article. Review.
-
Emerging role of non-coding RNAs in the course of HIV infection.Int Immunopharmacol. 2022 Feb;103:108460. doi: 10.1016/j.intimp.2021.108460. Epub 2021 Dec 20. Int Immunopharmacol. 2022. PMID: 34942460 Review.
Cited by
-
MicroRNA Profiling of the Inflammatory Response after Early and Late Asthmatic Reaction.Int J Mol Sci. 2024 Jan 22;25(2):1356. doi: 10.3390/ijms25021356. Int J Mol Sci. 2024. PMID: 38279356 Free PMC article.
-
RNA sequencing and bioinformatics analysis of circular RNAs in asphyxial newborns with acute kidney injury.Kaohsiung J Med Sci. 2023 Apr;39(4):337-344. doi: 10.1002/kjm2.12644. Epub 2023 Jan 19. Kaohsiung J Med Sci. 2023. PMID: 36655871 Free PMC article.
-
Patterns of Peripheral Blood B-Cell Subtypes Are Associated With Treatment Response in Patients Treated With Immune Checkpoint Inhibitors: A Prospective Longitudinal Pan-Cancer Study.Front Immunol. 2022 Apr 1;13:840207. doi: 10.3389/fimmu.2022.840207. eCollection 2022. Front Immunol. 2022. PMID: 35432362 Free PMC article.
-
Editorial: CD4+ T cells in HIV: A Friend or a Foe?Front Immunol. 2023 Jul 11;14:1203531. doi: 10.3389/fimmu.2023.1203531. eCollection 2023. Front Immunol. 2023. PMID: 37497218 Free PMC article. No abstract available.
-
Regulation of the tumor immune microenvironment by cancer-derived circular RNAs.Cell Death Dis. 2023 Feb 16;14(2):132. doi: 10.1038/s41419-023-05647-w. Cell Death Dis. 2023. PMID: 36797245 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

