Short-Term Steroid Treatment of Rhesus Macaque Increases Transduction
- PMID: 34806411
- PMCID: PMC8885436
- DOI: 10.1089/hum.2021.239
Short-Term Steroid Treatment of Rhesus Macaque Increases Transduction
Abstract
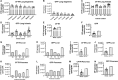
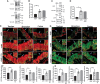
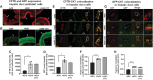
Repeat dosing poses a major hurdle for the development of an adeno-associated virus (AAV)-based gene therapy for cystic fibrosis, in part because of the potential for development of an immune reaction to the AAV1 capsid proteins. Here, to dampen the immune response to AAV1, we treated Rhesus monkeys with methylprednisolone before and after the instillation of two doses of AAV1Δ27-264-CFTR into their airways at 0 and 30 days, followed by a single dose of AAV1-GFP on day 60. Animals were euthanized on day 90, except for one monkey that was sacrificed at 1 year. No adverse events occurred, indicating that the two AAV1 vectors are safe. rAAV1-CFTR and AAV1-GFP vector genomes and mRNA transcripts were detectable in all lung sections and in the liver and pancreas at day 90 and after 1 year at levels comparable with animals necropsied at 90 days. The numbers of vector genomes for cystic fibrosis transmembrane regulator (CFTR) and green fluorescent protein (GFP) detected here were higher than those found in the monkeys infected without methylprednisolone treatment that we tested previously.1 Also, lung surface and keratin 5-positive basal cells showed higher CFTR and GFP staining than did the cells from the uninfected monkey control. Positive immunostaining, also detected in the liver and pancreas, remained stable for at least a year. All animals seroconverted for anticapsid antibodies by 90 days post-treatment. The neutralizing antibody titer declined in the animal necropsied at 1 year. Conclusion: AAV1 safely and effectively transduces monkey airway and basal cells. Both the presence of vector genomes and transduction from AAV1-CFTR and AAV1-GFP virus seen in the monkeys 4 months to 1 year after the first instillation suggest that repeat dosing with AAV1-based vectors is achievable, particularly after methylprednisolone treatment.
Keywords: AAV1; CFTR; Rhesus; basal cells; cystic fibrosis; lung liver; pancreas; repeat dosing; surface cells.
Conflict of interest statement
L.C. has a license agreement with RA Capital, and W.B.G. is a consultant for Vertex and Sarepta Pharmaceuticals in areas that are unrelated to this study.
Figures



Similar articles
-
Transduction of Surface and Basal Cells in Rhesus Macaque Lung Following Repeat Dosing with AAV1CFTR.Hum Gene Ther. 2020 Sep;31(17-18):1010-1023. doi: 10.1089/hum.2020.207. Hum Gene Ther. 2020. PMID: 32862701 Free PMC article.
-
Transduction of Ferret Surface and Basal Cells of Airways, Lung, Liver, and Pancreas via Intratracheal or Intravenous Delivery of Adeno-Associated Virus 1 or 6.Hum Gene Ther. 2023 Nov;34(21-22):1135-1144. doi: 10.1089/hum.2023.095. Epub 2023 Oct 16. Hum Gene Ther. 2023. PMID: 37650819 Free PMC article.
-
A Preclinical Study in Rhesus Macaques for Cystic Fibrosis to Assess Gene Transfer and Transduction by AAV1 and AAV5 with a Dual-Luciferase Reporter System.Hum Gene Ther Clin Dev. 2017 Sep;28(3):145-156. doi: 10.1089/humc.2017.067. Epub 2017 Jul 19. Hum Gene Ther Clin Dev. 2017. PMID: 28726496 Free PMC article.
-
Gene therapy in cystic fibrosis.Chest. 2001 Sep;120(3 Suppl):124S-131S. doi: 10.1378/chest.120.3_suppl.124s. Chest. 2001. PMID: 11555567 Review.
-
Recombinant adeno-associated virus vectors for cystic fibrosis gene therapy.Curr Opin Mol Ther. 2001 Oct;3(5):497-502. Curr Opin Mol Ther. 2001. PMID: 11699895 Review.
Cited by
-
AAV1-CFTR preferentially transduces cysts and reduces cyst size in a mouse model of ADPKD.Am J Physiol Cell Physiol. 2025 Jun 1;328(6):C1783-C1792. doi: 10.1152/ajpcell.00057.2025. Epub 2025 Apr 16. Am J Physiol Cell Physiol. 2025. PMID: 40237039 Free PMC article.
-
Gene therapy for cystic fibrosis: Challenges and prospects.Front Pharmacol. 2022 Oct 11;13:1015926. doi: 10.3389/fphar.2022.1015926. eCollection 2022. Front Pharmacol. 2022. PMID: 36304167 Free PMC article. Review.
-
Amelioration of airway and GI disease in G551D-CF ferrets by AAV1 and AAV6.Gene Ther. 2024 Sep;31(9-10):499-510. doi: 10.1038/s41434-024-00469-7. Epub 2024 Jul 28. Gene Ther. 2024. PMID: 39069560
References
-
- Ma CC, Wang ZL, Xu T, et al. . The approved gene therapy drugs worldwide: from 1998 to 2019. Biotechnol Adv 2020;40:107502. - PubMed
-
- Bernal JL, Andrews N, Gower C, et al. Early effectiveness of COVID-19 vaccination with BNT162b2 mRNA vaccine and ChAdOx1 adenovirus vector vaccine on symptomatic disease, hospitalisations and mortality in older adults in England. MedRxiv 2021. [Epub ahead of print]; DOI: 10.1101/2021.03.01.21252652. - DOI
-
- Collins FS. Cystic fibrosis: molecular biology and theraputic implications. Science 1992;256:774–779. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

