Randomized Controlled Study of Tenofovir versus Lamivudine Followed by Tenofovir in Severe Exacerbation of Hepatitis B
- PMID: 34807763
- PMCID: PMC8846303
- DOI: 10.1128/AAC.01664-21
Randomized Controlled Study of Tenofovir versus Lamivudine Followed by Tenofovir in Severe Exacerbation of Hepatitis B
Abstract
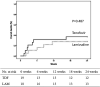
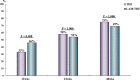
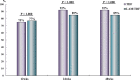
Spontaneous severe acute exacerbation (SAE) is not uncommon in the natural history of chronic hepatitis B (CHB). Lamivudine (LAM) has the advantages of low price, quick onset, good efficacy, and no drug resistance within 24 weeks. This study aimed to compare the short-term efficacy of tenofovir disoproxil fumarate (TDF) and LAM for 24 weeks followed by TDF in the treatment of CHB with severe acute exacerbation. Consecutive patients of CHB with SAE were randomized to receive either TDF (19 patients) or LAM for 24 weeks, followed by TDF (18 patients). The primary endpoint was overall mortality or receipt of liver transplantation by week 24. This study was approved by the Institutional Review Board (IRB) of the Kaohsiung Veterans General Hospital (VGHKS12-CT5-10). The baseline characteristics were comparable between the two groups. By week 24, seven (37%) and five (28%) patients in the TDF and LAM-TDF groups died or received liver transplantation (P = 0.487). Multivariate analysis showed that albumin level, prothrombin time (PT), and hepatic encephalopathy were independent factors associated with mortality or liver transplantation by week 24. Early reductions in HBV DNA of more than or equal to 2 log at 1 and 2 weeks were similar between the two groups. The biochemical and virological responses at 12, 24, and 48 weeks were also similar between the two groups. TDF and LAM for 24 weeks followed by TDF achieved a similar clinical outcome in CHB patients with SAE. (This study has been registered at ClinicalTrials.gov under identifier NCT01848743).
Keywords: acute exacerbation; exacerbation; hepatitis B virus; lamivudine; tenofovir disoproxil fumarate.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Tenofovir disoproxil fumarate is superior to lamivudine plus adefovir in lamivudine-resistant chronic hepatitis B patients.World J Gastroenterol. 2015 Mar 7;21(9):2746-53. doi: 10.3748/wjg.v21.i9.2746. World J Gastroenterol. 2015. PMID: 25759545 Free PMC article.
-
Tenofovir vs lamivudine plus adefovir in chronic hepatitis B: TENOSIMP-B study.World J Gastroenterol. 2017 Nov 7;23(41):7459-7469. doi: 10.3748/wjg.v23.i41.7459. World J Gastroenterol. 2017. PMID: 29151700 Free PMC article. Clinical Trial.
-
Comparison of tenofovir plus lamivudine versus tenofovir monotherapy in patients with lamivudine-resistant chronic hepatitis B.Clin Mol Hepatol. 2016 Mar;22(1):152-9. doi: 10.3350/cmh.2016.22.1.152. Epub 2016 Mar 28. Clin Mol Hepatol. 2016. PMID: 27044766 Free PMC article.
-
Efficacy of tenofovir-based rescue therapy in patients with lamivudine-resistant hepatitis B virus: A systematic review and meta-analysis.Clin Res Hepatol Gastroenterol. 2016 Sep;40(4):447-56. doi: 10.1016/j.clinre.2015.10.005. Epub 2015 Nov 24. Clin Res Hepatol Gastroenterol. 2016. PMID: 26621535
-
Development of tenofovir disoproxil fumarate resistance after complete viral suppression in a patient with treatment-naïve chronic hepatitis B: A case report and review of the literature.World J Gastroenterol. 2018 May 7;24(17):1919-1924. doi: 10.3748/wjg.v24.i17.1919. World J Gastroenterol. 2018. PMID: 29740207 Free PMC article. Review.
References
-
- Liaw YF. 1995. Acute exacerbation and superinfection in patients with chronic viral hepatitis. J Formos Med Assoc 94:521–528. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

