Prognostic impact of Schlafen 11 in bladder cancer patients treated with platinum-based chemotherapy
- PMID: 34808009
- PMCID: PMC8819307
- DOI: 10.1111/cas.15207
Prognostic impact of Schlafen 11 in bladder cancer patients treated with platinum-based chemotherapy
Abstract
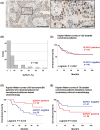
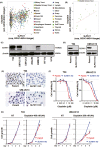
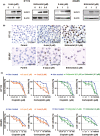
The utility of Schlafen 11 (SLFN11) expression as a predictive biomarker for platinum-based chemotherapy has been established for cancers from different histologies. However, the therapeutic relevance of SLFN11 in bladder cancer (BC) is unknown. Here, we examined the clinicopathologic significance of SLFN11 expression across 120 BC cases by immunohistochemistry. We divided the cases into two cohorts, one including 50 patients who received adjuvant or neoadjuvant platinum-based chemotherapy, and the other including 70 BC patients treated by surgical resection without chemotherapy. In the cohort of 50 BC cases treated with platinum-based chemotherapy, the SLFN11-positive group (n = 25) showed significantly better overall survival than the SLFN11-negative group (n = 25, P = .012). Schlafen 11 expression correlated significantly with the expression of luminal subtype marker GATA3. Multivariate analyses identified SLFN11 expression as an independent prognostic predictor (odds ratio, 0.32; 95% confidence interval, 0.11-0.91; P = .033). Conversely, in the cohort of 70 BC cases not receiving platinum-based chemotherapy, the SLFN11-positive group (n = 29) showed significantly worse overall survival than the SLFN11-negative group (n = 41, P = .034). In vitro analyses using multiple BC cell lines confirmed that SLFN11 KO rendered cells resistant to cisplatin. The epigenetic modifying drugs 5-azacytidine and entinostat restored SLFN11 expression and resensitized cells to cisplatin and carboplatin in SLFN11-negative BC cell lines. We conclude that SLFN11 is a predictive biomarker for BC patients who undergo platinum-based chemotherapy and that the combination of epigenetic modifiers could rescue refractory BC patients to platinum derivatives by reactivating SLFN11 expression.
Keywords: SLFN11; biomarker; bladder cancer; chemotherapy; cisplatin.
© 2021 The Authors. Cancer Science published by John Wiley & Sons Australia, Ltd on behalf of Japanese Cancer Association.
Figures

Similar articles
-
SLFN11 Expression in Advanced Prostate Cancer and Response to Platinum-based Chemotherapy.Mol Cancer Ther. 2020 May;19(5):1157-1164. doi: 10.1158/1535-7163.MCT-19-0926. Epub 2020 Mar 3. Mol Cancer Ther. 2020. PMID: 32127465 Free PMC article.
-
Schlafen 11 predicts response to platinum-based chemotherapy in gastric cancers.Br J Cancer. 2021 Jul;125(1):65-77. doi: 10.1038/s41416-021-01364-3. Epub 2021 Mar 30. Br J Cancer. 2021. PMID: 33785877 Free PMC article.
-
Epigenetic inactivation of the putative DNA/RNA helicase SLFN11 in human cancer confers resistance to platinum drugs.Oncotarget. 2016 Jan 19;7(3):3084-97. doi: 10.18632/oncotarget.6413. Oncotarget. 2016. PMID: 26625211 Free PMC article.
-
Schlafen 11 (SLFN11), a restriction factor for replicative stress induced by DNA-targeting anti-cancer therapies.Pharmacol Ther. 2019 Sep;201:94-102. doi: 10.1016/j.pharmthera.2019.05.009. Epub 2019 May 23. Pharmacol Ther. 2019. PMID: 31128155 Free PMC article. Review.
-
From predictive biomarker to therapeutic target: the dual role of SLFN11 in chemotherapy sensitivity.Cancer Chemother Pharmacol. 2025 Jun 18;95(1):60. doi: 10.1007/s00280-025-04781-w. Cancer Chemother Pharmacol. 2025. PMID: 40531330 Review.
Cited by
-
Can Schlafen 11 Help to Stratify Ovarian Cancer Patients Treated with DNA-Damaging Agents?Cancers (Basel). 2022 May 10;14(10):2353. doi: 10.3390/cancers14102353. Cancers (Basel). 2022. PMID: 35625957 Free PMC article. Review.
-
Schlafens: Emerging Therapeutic Targets.Cancers (Basel). 2024 May 9;16(10):1805. doi: 10.3390/cancers16101805. Cancers (Basel). 2024. PMID: 38791884 Free PMC article. Review.
-
Schlafen 11 (SLFN11) Kills Cancer Cells Undergoing Unscheduled Re-replication.Mol Cancer Ther. 2023 Aug 1;22(8):985-995. doi: 10.1158/1535-7163.MCT-22-0552. Mol Cancer Ther. 2023. PMID: 37216280 Free PMC article.
-
Reconsidering the mechanisms of action of PARP inhibitors based on clinical outcomes.Cancer Sci. 2022 Sep;113(9):2943-2951. doi: 10.1111/cas.15477. Epub 2022 Jul 16. Cancer Sci. 2022. PMID: 35766436 Free PMC article. Review.
-
Protein-Based Predictive Biomarkers to Personalize Neoadjuvant Therapy for Bladder Cancer-A Systematic Review of the Current Status.Int J Mol Sci. 2024 Sep 13;25(18):9899. doi: 10.3390/ijms25189899. Int J Mol Sci. 2024. PMID: 39337385 Free PMC article.
References
-
- Antoni S, Ferlay J, Soerjomataram I, Znaor A, Jemal A, Bray F. Bladder cancer incidence and mortality: a global overview and recent trends. Eur Urol. 2017;71:96‐108. - PubMed
-
- Smith AB, Deal AM, Woods ME, et al. Muscle‐invasive bladder cancer: evaluating treatment and survival in the National Cancer Data Base. BJU Int. 2014;114:719‐726. - PubMed
-
- Powles T, van der Heijden MS, Castellano D, et al. Durvalumab alone and durvalumab plus tremelimumab versus chemotherapy in previously untreated patients with unresectable, locally advanced or metastatic urothelial carcinoma (DANUBE): a randomised, open‐label, multicentre, phase 3 trial. Lancet Oncol. 2020;21:1574‐1588. - PubMed
-
- von der Maase H, Sengelov L, Roberts JT, et al. Long‐term survival results of a randomized trial comparing gemcitabine plus cisplatin, with methotrexate, vinblastine, doxorubicin, plus cisplatin in patients with bladder cancer. J Clin Oncol. 2005;23:4602‐4608. - PubMed
MeSH terms
Substances
Grants and funding
- Z01-BC-006150/Center for Cancer Research, the Intramural Program of the National Cancer Institute
- 26670175/Japan Society for the Promotion of Science
- JP15H04713/Japan Society for the Promotion of Science
- JP16H06999/Japan Society for the Promotion of Science
- JP16K08691/Japan Society for the Promotion of Science
LinkOut - more resources
Full Text Sources
Medical
Research Materials

