Risk of bias in observational studies using routinely collected data of comparative effectiveness research: a meta-research study
- PMID: 34809637
- PMCID: PMC8608432
- DOI: 10.1186/s12916-021-02151-w
Risk of bias in observational studies using routinely collected data of comparative effectiveness research: a meta-research study
Abstract
Background: To assess the completeness of reporting, research transparency practices, and risk of selection and immortal bias in observational studies using routinely collected data for comparative effectiveness research.
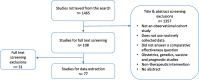
Method: We performed a meta-research study by searching PubMed for comparative effectiveness observational studies evaluating therapeutic interventions using routinely collected data published in high impact factor journals from 01/06/2018 to 30/06/2020. We assessed the reporting of the study design (i.e., eligibility, treatment assignment, and the start of follow-up). The risk of selection bias and immortal time bias was determined by assessing if the time of eligibility, the treatment assignment, and the start of follow-up were synchronized to mimic the randomization following the target trial emulation framework.
Result: Seventy-seven articles were identified. Most studies evaluated pharmacological treatments (69%) with a median sample size of 24,000 individuals. In total, 20% of articles inadequately reported essential information of the study design. One-third of the articles (n = 25, 33%) raised some concerns because of unclear reporting (n = 6, 8%) or were at high risk of selection bias and/or immortal time bias (n = 19, 25%). Only five articles (25%) described a solution to mitigate these biases. Six articles (31%) discussed these biases in the limitations section.
Conclusion: Reporting of essential information of study design in observational studies remained suboptimal. Selection bias and immortal time bias were common methodological issues that researchers and physicians should be aware of when interpreting the results of observational studies using routinely collected data.
Keywords: Emulated trial; Meta-research; Observational studies; Risk of bias; Routinely collected data.
© 2021. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

Similar articles
-
Specifying a target trial prevents immortal time bias and other self-inflicted injuries in observational analyses.J Clin Epidemiol. 2016 Nov;79:70-75. doi: 10.1016/j.jclinepi.2016.04.014. Epub 2016 May 27. J Clin Epidemiol. 2016. PMID: 27237061 Free PMC article. Review.
-
Conducting observational analyses with the target trial emulation approach: a methodological systematic review.BMJ Open. 2024 Nov 12;14(11):e086595. doi: 10.1136/bmjopen-2024-086595. BMJ Open. 2024. PMID: 39532374 Free PMC article.
-
Methodological biases in observational hospital studies of COVID-19 treatment effectiveness: pitfalls and potential.Front Med (Lausanne). 2024 Mar 21;11:1362192. doi: 10.3389/fmed.2024.1362192. eCollection 2024. Front Med (Lausanne). 2024. PMID: 38576716 Free PMC article.
-
Improving rheumatoid arthritis comparative effectiveness research through causal inference principles: systematic review using a target trial emulation framework.Ann Rheum Dis. 2020 Jul;79(7):883-890. doi: 10.1136/annrheumdis-2020-217200. Epub 2020 May 7. Ann Rheum Dis. 2020. PMID: 32381560 Free PMC article.
-
Implementation of the trial emulation approach in medical research: a scoping review.BMC Med Res Methodol. 2023 Aug 16;23(1):186. doi: 10.1186/s12874-023-02000-9. BMC Med Res Methodol. 2023. PMID: 37587484 Free PMC article.
Cited by
-
Reporting of Observational Studies Explicitly Aiming to Emulate Randomized Trials: A Systematic Review.JAMA Netw Open. 2023 Sep 5;6(9):e2336023. doi: 10.1001/jamanetworkopen.2023.36023. JAMA Netw Open. 2023. PMID: 37755828 Free PMC article.
-
"In conferences, everyone goes 'health data is the future' ": an interview study on challenges in re-using EHR data for research in Clinical Data Warehouses.AMIA Annu Symp Proc. 2024 Jan 11;2023:579-588. eCollection 2023. AMIA Annu Symp Proc. 2024. PMID: 38222365 Free PMC article.
-
Exploring the causal effect of omega-3 polyunsaturated fatty acid levels on the risk of type 1 diabetes: a Mendelian randomization study.Front Genet. 2024 Jul 8;15:1353081. doi: 10.3389/fgene.2024.1353081. eCollection 2024. Front Genet. 2024. PMID: 39040994 Free PMC article.
-
Outcomes after early treatment with hydroxychloroquine and azithromycin: An analysis of a database of 30,423 COVID-19 patients.New Microbes New Infect. 2023 Oct 30;55:101188. doi: 10.1016/j.nmni.2023.101188. eCollection 2023 Oct. New Microbes New Infect. 2023. PMID: 38024333 Free PMC article.
-
Causal relationship between dietary factors and breast cancer risk: A Mendelian randomization study.Heliyon. 2023 Oct 14;9(10):e20980. doi: 10.1016/j.heliyon.2023.e20980. eCollection 2023 Oct. Heliyon. 2023. PMID: 37867896 Free PMC article.
References
-
- Hernan M, Robins JM. Causal inference: what if. Boca Raton: Chapman & Hall/CRC; 2020.
MeSH terms
LinkOut - more resources
Full Text Sources

