HFIP Mediates a Direct C-C Coupling between Michael Acceptors and Eschenmoser's salt
- PMID: 34813137
- PMCID: PMC9306631
- DOI: 10.1002/anie.202109933
HFIP Mediates a Direct C-C Coupling between Michael Acceptors and Eschenmoser's salt
Abstract
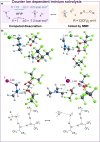
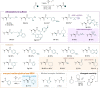
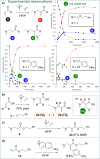
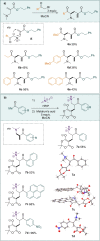
A direct C-C coupling process that merges Michael acceptors and Eschenmoser's salt is presented. Although reminiscent of the Morita-Baylis-Hillman reaction, this process requires no Lewis base catalyst. The underlying mechanism was unveiled by a combination of kinetic, isotopic labelling experiments as well as computational investigations, which showcased the critical role of HFIP as a superior mediator for proton-transfer events as well as the decisive role of the halide counterion.
Keywords: Amines; Hydrogen bonds; Iodine; Quantum chemistry; Solvent effects.
© 2021 The Authors. Angewandte Chemie International Edition published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- None
-
- Morita K., Suzuki Z., Hirose H., Bull. Chem. Soc. Jpn. 1968, 41, 2815–2815;
-
- Hillman M. E. D., Baylis A. B., German Patent 2155113, 1972.
-
- None
-
- Ciganek E. in Organic Reactions, Vol. 51 (Ed.: Paquette L. A.), Wiley, New York, 1997, pp. 201–350;
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

