Comparison of the Performance of the PanBio COVID-19 Antigen Test in SARS-CoV-2 B.1.1.7 (Alpha) Variants versus non-B.1.1.7 Variants
- PMID: 34817226
- PMCID: PMC8612141
- DOI: 10.1128/Spectrum.00884-21
Comparison of the Performance of the PanBio COVID-19 Antigen Test in SARS-CoV-2 B.1.1.7 (Alpha) Variants versus non-B.1.1.7 Variants
Abstract
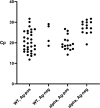
This study evaluates the performance of the PanBio COVID-19 antigen (Ag) test as part of a hospital infection control policy. Hospital staff was encouraged to get tested for COVID-19 when presenting with SARS-CoV-2-related symptoms. In a period of approximately 5 months, a steady decline in the performance of the Ag test was noted, epidemiologically coinciding with the rise of the SARS-CoV-2 B.1.1.7 (alpha) variant of concern (VOC) in the Netherlands. This led to the hypothesis that the diagnostic performance of the PanBio COVID-19 Ag test was influenced by the infecting viral variant. The results show a significantly lower sensitivity of the PanBio COVID-19 Ag test in persons infected with the B.1.1.7 (alpha) variant of SARS-CoV-2 in comparison with that in persons infected with non-B.1.1.7 variants, also after adjustment for viral load. IMPORTANCE Antigen tests for COVID-19 are widely used for rapid identification of COVID-19 cases, for example, for access to schools, festivals, and travel. There are several FDA- and CE-cleared tests on the market. Their performance has been evaluated mainly on the basis of infections by the classical variant of the causing virus, SARS-CoV-2. This paper provides evidence that the performance of one of the most widely used antigen tests detects significantly fewer cases of COVID-19 by the alpha variant than by the classical variants of SARS-CoV-2. This means that the role of antigen tests needs to be reevaluated in regions where other variants of SARS-CoV-2 predominate.
Keywords: B.1.1.7 variant; COVID-19; SARS-CoV-2; antigen; diagnostics.
Conflict of interest statement
We declare no conflicts of interest.
Figures
References
-
- Dinnes J, Deeks JJ, Berhane S, Taylor M, Adriano A, Davenport C, Dittrich S, Emperador D, Takwoingi Y, Cunningham J, Beese S, Domen J, Dretzke J, Ferrante di Ruffano L, Harris IM, Price MJ, Taylor-Phillips S, Hooft L, Leeflang MMG, McInnes MDF, Spijker R, Van den Bruel A, Cochrane COVID-19 Diagnostic Test Accuracy Group. 2021. Rapid, point-of-care antigen and molecular-based tests for diagnosis of SARS-CoV-2 infection. Cochrane Database Syst Rev 3:CD013705. doi:10.1002/14651858.CD013705.pub2. - DOI - PMC - PubMed
-
- Gremmels H, Winkel BMF, Schuurman R, Rosingh A, Rigter NAM, Rodriguez O, Ubijaan J, Wensing AMJ, Bonten MJM, Hofstra LM. 2021. Real-life validation of the Panbio COVID19 antigen rapid test (Abbott) in community-dwelling subjects with symptoms of potential SARS-CoV-2 infection. E Clinical Medicine 31:100677. doi:10.1016/j.eclinm.2020.100677. - DOI - PMC - PubMed
-
- Voss C, Esmail S, Liu X, Knauer MJ, Ackloo S, Kaneko T, Lowes L, Stogios P, Seitova A, Hutchinson A, Yusifov F, Skarina T, Evdokimova E, Loppnau P, Ghiabi P, Haijan T, Zhong S, Abdoh H, Hedley BD, Bhayana V, Martin CM, Slessarev M, Chin-Yee B, Fraser DD, Chin-Yee I, Li SS. 2021. Epitope-specific antibody responses differentiate COVID-19 outcomes and variants of concern. JCI Insight 6:e148855. - PMC - PubMed
-
- Singh J, Samal J, Kumar V, Sharma J, Agrawal U, Ehtesham NZ, Sundar D, Rahman SA, Hira S, Hasnain SE. 2021. Structure-function analyses of new SARS-CoV-2 variants B.1.1.7, B.1.351 and B.1.1.28.1: clinical, diagnostic, therapeutic and public health implications. Viruses 13:439. doi:10.3390/v13030439. - DOI - PMC - PubMed
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous