Novel chicken two-dimensional intestinal model comprising all key epithelial cell types and a mesenchymal sub-layer
- PMID: 34819162
- PMCID: PMC8611946
- DOI: 10.1186/s13567-021-01010-z
Novel chicken two-dimensional intestinal model comprising all key epithelial cell types and a mesenchymal sub-layer
Abstract
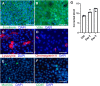
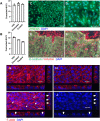
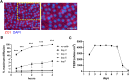
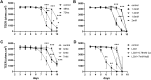
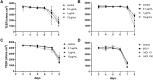
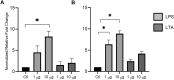
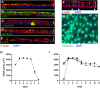
The intestinal epithelium plays a variety of roles including providing an effective physical barrier and innate immune protection against infection. Two-dimensional models of the intestinal epithelium, 2D enteroids, are a valuable resource to investigate intestinal cell biology and innate immune functions and are suitable for high throughput studies of paracellular transport and epithelial integrity. We have developed a chicken 2D enteroid model that recapitulates all major differentiated cell lineages, including enterocytes, Paneth cells, Goblet cells, enteroendocrine cells and leukocytes, and self-organises into an epithelial and mesenchymal sub-layer. Functional studies demonstrated the 2D enteroids formed a tight cell layer with minimal paracellular flux and a robust epithelial integrity, which was maintained or rescued following damage. The 2D enteroids were also able to demonstrate appropriate innate immune responses following exposure to bacterial endotoxins, from Salmonella enterica serotype Typhimurium and Bacillus subtilis. Frozen 2D enteroids cells when thawed were comparable to freshly isolated cells. The chicken 2D enteroids provide a useful ex vivo model to study intestinal cell biology and innate immune function, and have potential uses in screening of nutritional supplements, pharmaceuticals, and bioactive compounds.
Keywords: 2D enteroid; Bacteria; Barrier integrity; Chicken; Gut model; Immune response; Intestinal epithelium; Organoid.
© 2021. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

Similar articles
-
Disentangling the innate immune responses of intestinal epithelial cells and lamina propria cells to Salmonella Typhimurium infection in chickens.Front Microbiol. 2023 Oct 3;14:1258796. doi: 10.3389/fmicb.2023.1258796. eCollection 2023. Front Microbiol. 2023. PMID: 37854334 Free PMC article.
-
Infection of porcine enteroids and 2D differentiated intestinal epithelial cells with rotavirus A to study cell tropism and polarized immune response.Emerg Microbes Infect. 2023 Dec;12(2):2239937. doi: 10.1080/22221751.2023.2239937. Emerg Microbes Infect. 2023. PMID: 37483148 Free PMC article.
-
Salmonella enterica Serovar Typhimurium Exploits Cycling through Epithelial Cells To Colonize Human and Murine Enteroids.mBio. 2021 Jan 12;12(1):e02684-20. doi: 10.1128/mBio.02684-20. mBio. 2021. PMID: 33436434 Free PMC article.
-
Enteroids for Nutritional Studies.Mol Nutr Food Res. 2019 Aug;63(16):e1801143. doi: 10.1002/mnfr.201801143. Epub 2019 Apr 10. Mol Nutr Food Res. 2019. PMID: 30883003 Review.
-
Enteroids: Promising in Vitro Models for Studies of Intestinal Physiology and Nutrition in Farm Animals.J Agric Food Chem. 2019 Mar 6;67(9):2421-2428. doi: 10.1021/acs.jafc.8b06908. Epub 2019 Feb 19. J Agric Food Chem. 2019. PMID: 30739438 Review.
Cited by
-
3D sheep rumen epithelial structures driven from single cells in vitro.Vet Res. 2023 Nov 9;54(1):104. doi: 10.1186/s13567-023-01234-1. Vet Res. 2023. PMID: 37946298 Free PMC article.
-
Long-term culture of chicken tracheal organoids for the purpose of avian influenza virus research.Virol J. 2025 Apr 15;22(1):99. doi: 10.1186/s12985-025-02714-w. Virol J. 2025. PMID: 40234888 Free PMC article.
-
Discrimination of distinct chicken M cell subsets based on CSF1R expression.Sci Rep. 2024 Apr 16;14(1):8795. doi: 10.1038/s41598-024-59368-x. Sci Rep. 2024. PMID: 38627516 Free PMC article.
-
Disentangling the innate immune responses of intestinal epithelial cells and lamina propria cells to Salmonella Typhimurium infection in chickens.Front Microbiol. 2023 Oct 3;14:1258796. doi: 10.3389/fmicb.2023.1258796. eCollection 2023. Front Microbiol. 2023. PMID: 37854334 Free PMC article.
-
Differential Salmonella Typhimurium intracellular replication and host cell responses in caecal and ileal organoids derived from chicken.Vet Res. 2023 Jul 31;54(1):63. doi: 10.1186/s13567-023-01189-3. Vet Res. 2023. PMID: 37525204 Free PMC article.
References
-
- Peterson LW, Artis D. Intestinal epithelial cells: regulators of barrier function and immune homeostasis. Nat Rev Immunol. 2014;14:141–153. - PubMed
-
- Roulis M, Flavell RA. Fibroblasts and myofibroblasts of the intestinal lamina propria in physiology and disease. Differentiation. 2016;92:116–131. - PubMed
-
- Fujii M, Matano M, Toshimitsu K, Takano A, Mikami Y, Nishikori S, Sugimoto S, Sato T. Human intestinal organoids maintain self-renewal capacity and cellular diversity in niche-inspired culture condition. Cell Stem Cell. 2018;23:787–793.e786. - PubMed
-
- Sato T, Stange DE, Ferrante M, Vries RGJ, van Es JH, van den Brink S, van Houdt WJ, Pronk A, van Gorp J, Siersema PD, Clevers H. Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterology. 2011;141:1762–1772. - PubMed
-
- Sato T, Vries RG, Snippert HJ, van de Wetering M, Barker N, Stange DE, van Es JH, Abo A, Kujala P, Peters PJ, Clevers H. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature. 2009;459:262–265. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

