E7-Targeted Nanotherapeutics for Key HPV Afflicted Cervical Lesions by Employing CRISPR/Cas9 and Poly (Beta-Amino Ester)
- PMID: 34819726
- PMCID: PMC8606985
- DOI: 10.2147/IJN.S335277
E7-Targeted Nanotherapeutics for Key HPV Afflicted Cervical Lesions by Employing CRISPR/Cas9 and Poly (Beta-Amino Ester)
Abstract
Introduction: Persistent HR-HPV (high-risk human papillomavirus) infection is the main cause of cervical cancer. The HPV oncogene E7 plays a key role in HPV tumorigenesis. At present, HPV preventive vaccines are not effective for patients who already have a cervical disease, and implementation of the recommended regular cervical screening is difficult in countries and regions lacking medical resources. Therefore, patients need medications to treat existing HPV infections and thus block the progression of cervical disease.
Methods: In this study, we developed nanoparticles (NPs) composed of the non-viral vector PBAE546 and a CRISPR/Cas9 recombinant plasmid targeting HPV16 E7 as a vaginal treatment for HPV infection and related cervical malignancies.
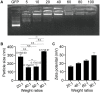
Results: Our NPs showed low toxicity and high biological safety both in vitro (cell line viability) and in vivo (various important organs of mice). Our NPs significantly inhibited the growth of xenograft tumors derived from cervical cancer cell lines in nude mice and significantly reversed the cervical epithelial malignant phenotype of HPV16 transgenic mice.
Conclusion: Our NPs have great potential to be developed as a drug for the treatment of HPV-related cervical cancer and precancerous lesions.
Keywords: HPV16 transgenic mice; HR-HPV infection; PBAE; cervical cancer; gene therapy; nanoparticles.
© 2021 Xiong et al.
Conflict of interest statement
The authors report no potential conflicts of interest in this work.
Figures






Similar articles
-
Hyperbranched poly(β-amino ester) based polyplex nanopaticles for delivery of CRISPR/Cas9 system and treatment of HPV infection associated cervical cancer.J Control Release. 2020 May 10;321:654-668. doi: 10.1016/j.jconrel.2020.02.045. Epub 2020 Feb 28. J Control Release. 2020. PMID: 32114092
-
Nanoparticles Based on Poly (β-Amino Ester) and HPV16-Targeting CRISPR/shRNA as Potential Drugs for HPV16-Related Cervical Malignancy.Mol Ther. 2018 Oct 3;26(10):2443-2455. doi: 10.1016/j.ymthe.2018.07.019. Epub 2018 Jul 25. Mol Ther. 2018. PMID: 30241742 Free PMC article.
-
The application of CRISPR/Cas9 system in cervical carcinogenesis.Cancer Gene Ther. 2022 May;29(5):466-474. doi: 10.1038/s41417-021-00366-w. Epub 2021 Aug 4. Cancer Gene Ther. 2022. PMID: 34349239 Free PMC article.
-
In vitro and in vivo growth inhibition of human cervical cancer cells via human papillomavirus E6/E7 mRNAs' cleavage by CRISPR/Cas13a system.Antiviral Res. 2020 Jun;178:104794. doi: 10.1016/j.antiviral.2020.104794. Epub 2020 Apr 14. Antiviral Res. 2020. PMID: 32298665
-
Current and future direction in treatment of HPV-related cervical disease.J Mol Med (Berl). 2022 Jun;100(6):829-845. doi: 10.1007/s00109-022-02199-y. Epub 2022 Apr 27. J Mol Med (Berl). 2022. PMID: 35478255 Free PMC article. Review.
Cited by
-
Description of CRISPR-Cas9 development and its prospects in human papillomavirus-driven cancer treatment.Front Immunol. 2022 Nov 21;13:1037124. doi: 10.3389/fimmu.2022.1037124. eCollection 2022. Front Immunol. 2022. PMID: 36479105 Free PMC article. Review.
-
Gene Therapy for Malignant and Benign Gynaecological Disorders: A Systematic Review of an Emerging Success Story.Cancers (Basel). 2022 Jun 30;14(13):3238. doi: 10.3390/cancers14133238. Cancers (Basel). 2022. PMID: 35805007 Free PMC article. Review.
-
Human papillomavirus associated cervical lesion: pathogenesis and therapeutic interventions.MedComm (2020). 2023 Sep 14;4(5):e368. doi: 10.1002/mco2.368. eCollection 2023 Oct. MedComm (2020). 2023. PMID: 37719443 Free PMC article. Review.
-
Towards the elimination of infectious HPV: exploiting CRISPR/Cas innovations.Front Cell Infect Microbiol. 2025 Aug 4;15:1627668. doi: 10.3389/fcimb.2025.1627668. eCollection 2025. Front Cell Infect Microbiol. 2025. PMID: 40831701 Free PMC article. Review.
-
Bioinformatic and literature assessment of toxicity and allergenicity of a CRISPR-Cas9 engineered gene drive to control Anopheles gambiae the mosquito vector of human malaria.Malar J. 2023 Aug 14;22(1):234. doi: 10.1186/s12936-023-04665-5. Malar J. 2023. PMID: 37580703 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

