Nociceptin/orphanin FQ opioid receptor (NOP) selective ligand MCOPPB links anxiolytic and senolytic effects
- PMID: 34820764
- PMCID: PMC8612119
- DOI: 10.1007/s11357-021-00487-y
Nociceptin/orphanin FQ opioid receptor (NOP) selective ligand MCOPPB links anxiolytic and senolytic effects
Abstract
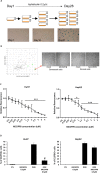
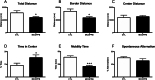
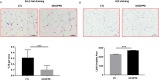
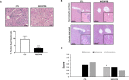
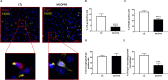
Accumulation of senescent cells may drive age-associated alterations and pathologies. Senolytics are promising therapeutics that can preferentially eliminate senescent cells. Here, we performed a high-throughput automatized screening (HTS) of the commercial LOPAC®Pfizer library on aphidicolin-induced senescent human fibroblasts, to identify novel senolytics. We discovered the nociceptin receptor FQ opioid receptor (NOP) selective ligand 1-[1-(1-methylcyclooctyl)-4-piperidinyl]-2-[(3R)-3-piperidinyl]-1H-benzimidazole (MCOPPB, a compound previously studied as potential anxiolytic) as the best scoring hit. The ability of MCOPPB to eliminate senescent cells in in vitro models was further tested in mice and in C. elegans. MCOPPB reduced the senescence cell burden in peripheral tissues but not in the central nervous system. Mice and worms exposed to MCOPPB also exhibited locomotion and lipid storage changes. Mechanistically, MCOPPB treatment activated transcriptional networks involved in the immune responses to external stressors, implicating Toll-like receptors (TLRs). Our study uncovers MCOPPB as a NOP ligand that, apart from anxiolytic effects, also shows tissue-specific senolytic effects.
Keywords: Aging; NOP; Senescence; Senolytic.
© 2021. The Author(s), under exclusive licence to American Aging Association.
Conflict of interest statement
MM, JB, and MH are co-inventors on a patent application: EP 3 552 605 A1, utilizing MCOPPB as a medicament with senolytic effects. Other authors declare no other competing interests.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

