Do Hydrothermal Shrimp Smell Vents?
- PMID: 34821843
- PMCID: PMC8623306
- DOI: 10.3390/insects12111043
Do Hydrothermal Shrimp Smell Vents?
Abstract
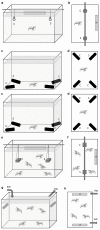
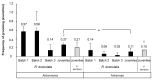
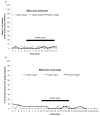
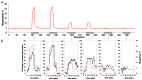
Deep-sea species endemic to hydrothermal vents face the critical challenge of detecting active sites in a vast environment devoid of sunlight. This certainly requires specific sensory abilities, among which olfaction could be a relevant sensory modality, since chemical compounds in hydrothermal fluids or food odors could potentially serve as orientation cues. The temperature of the vent fluid might also be used for locating vent sites. The objective of this study is to observe the following key behaviors of olfaction in hydrothermal shrimp, which could provide an insight into their olfactory capacities: (1) grooming behavior; (2) attraction to environmental cues (food odors and fluid markers). We designed experiments at both deep-sea and atmospheric pressure to assess the behavior of the vent shrimp Rimicaris exoculata and Mirocaris fortunata, as well as of the coastal species Palaemon elegans and Palaemon serratus for comparison. Here, we show that hydrothermal shrimp groom their sensory appendages similarly to other crustaceans, but this does not clean the dense bacterial biofilm that covers the olfactory structures. These shrimp have previously been shown to possess functional sensory structures, and to detect the environmental olfactory signals tested, but we do not observe significant attraction behavior here. Only temperature, as a signature of vent fluids, clearly attracts vent shrimp and thus is confirmed to be a relevant signal for orientation in their environment.
Keywords: antennules; behavior; chemosensory perception; grooming; hydrothermal shrimp; olfaction; thermal detection.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures









References
-
- Steullet P., Dudar O., Flavus T., Zhou M., Derby C.D. Selective ablation of antennular sensilla on the caribbean spiny lobster Panulirus argus suggests that dual antennular chemosensory pathways mediate odorant activation of searching and localization of food. J. Exp. Biol. 2001;204:4259–4269. doi: 10.1242/jeb.204.24.4259. - DOI - PubMed
-
- Derby C.D., Weissburg M.J. The chemical senses and chemosensory ecology of crustaceans. In: Derby C.D., Thiel M., editors. Crustacean Nervous Systems and Their Control of Behavior. Springer; New York, NY, USA: 2014. pp. 263–293.
-
- Schmidt M., Ache B.W. Processing of antennular input in the brain of the spiny lobster, Panulirus argus. J. Comp. Phys. A. 1996;178:605–628. doi: 10.1007/BF00227375. - DOI
-
- Ache B.W. Chemoreception and thermoreception. In: Bliss D., editor. The Biology of Crustacea. Volume 3. Academic Press; New York, NY, USA: 1982. pp. 369–398.
Grants and funding
LinkOut - more resources
Full Text Sources

