Gut microbiota depletion from early adolescence alters anxiety and depression-related behaviours in male mice with Alzheimer-like disease
- PMID: 34824332
- PMCID: PMC8617202
- DOI: 10.1038/s41598-021-02231-0
Gut microbiota depletion from early adolescence alters anxiety and depression-related behaviours in male mice with Alzheimer-like disease
Abstract
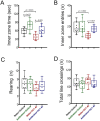
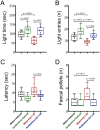
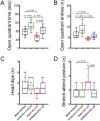
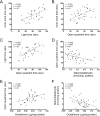
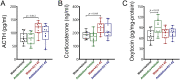
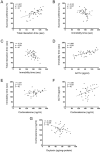
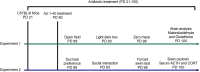
The gut-microbiota-brain axis plays an important role in stress-related disorders, and dysfunction of this complex bidirectional system is associated with Alzheimer's disease. This study aimed to assess the idea that whether gut microbiota depletion from early adolescence can alter anxiety- and depression-related behaviours in adult mice with or without Alzheimer-like disease. Male C57BL/6 mice were treated with an antibiotic cocktail from weaning to adulthood. Adult mice received an intracerebroventricular injection of amyloid-beta (Aβ)1-42, and were subjected to anxiety and depression tests. We measured, brain malondialdehyde and glutathione following anxiety tests, and assessed brain oxytocin and the hypothalamic-pituitary-adrenal (HPA) axis function by measuring adrenocorticotrophic hormone (ACTH) and corticosterone following depression tests. Healthy antibiotic-treated mice displayed significant decreases in anxiety-like behaviours, whereas they did not show any alterations in depression-like behaviours and HPA axis function. Antibiotic treatment from early adolescence prevented the development of anxiety- and depression-related behaviours, oxidative stress and HPA axis dysregulation in Alzheimer-induced mice. Antibiotic treatment increased oxytocin in the brain of healthy but not Alzheimer-induced mice. Taken together, these findings suggest that gut microbiota depletion following antibiotic treatment from early adolescence might profoundly affect anxiety- and depression-related behaviours, and HPA axis function in adult mice with Alzheimer-like disease.
© 2021. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


References
-
- Cryan, J. F. et al. The microbiota–gut–brain axis. Physiol. Rev.99, 1877–2013 (2019). - PubMed
-
- Sarkar SR, Banerjee S. Gut microbiota in neurodegenerative disorders. J. Neuroimmunol. 2019;328:98–104. - PubMed
-
- Luna RA, Foster JA. Gut brain axis: Diet microbiota interactions and implications for modulation of anxiety and depression. Curr. Opin. Biotechnol. 2015;32:35–41. - PubMed
-
- Zeraati, M., Enayati, M., Kafami, L., Shahidi, S. H. & Salari, A.-A. Gut microbiota depletion from early adolescence alters adult immunological and neurobehavioral responses in a mouse model of multiple sclerosis. Neuropharmacology157, 107685 (2019). - PubMed

